Understanding cholangiocyte biology
Cholangiopathy: a biliary disorder
Clinical breakthroughs against cholangiopathies
References
Further reading
The biliary system comprises extrahepatic bile ducts, intrahepatic bile ducts, and the gallbladder. The intrahepatic and extrahepatic biliary tree is lined with epithelial cells known as cholangiocytes. These epithelial cells are associated with vital physiologic processes that are crucial for normal body functioning.
Considering the importance of cholangiocytes in normal homeostasis of the biliary tree, the concept of cholangiopathy has emerged, which deals with a disease state due to the abnormal functioning of cholangiocytes. This article focuses on cholangiocyte biology and incidence of cholangiopathies.1
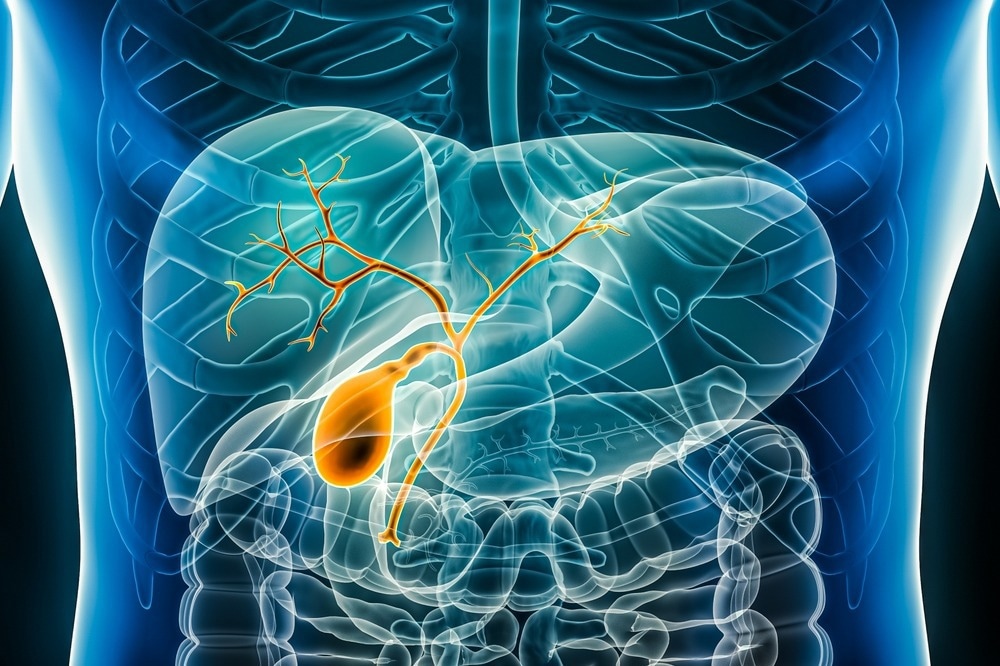
Image Credit: MattL_Images/Shutterstock.com
Understanding cholangiocyte biology
Bile is a yellow-greenish fluid secreted by the hepatocytes, a major parenchymal cell in the liver, that helps in food digestion. Bile is transported through the bile tract that is lined with cholangiocytes, covering around 2km in the human body. The primary function of epithelial cells, including cholangiocytes, is to control the molecular and ionic diffusion through the epithelial barrier selectively. However, in contrast to epithelial cells, cholangiocytes are functionally and morphologically heterogeneous.
A study of cholangiocytes under electron microscopy revealed the presence of apical microvilli that faces the lumen of the bile duct. In addition, the presence of various micro-organelles, such as mitochondria, the rough endoplasmic reticulum, the nucleus in the cytoplasm, and vesicles, were noted.2 This finding implies that cholangiocytes are extremely versatile and are associated with complex functions.
Cholangiocytes are differentiated through stem cells in the peribiliary glands. Small cholangiocytes have proliferative capabilities and exhibit functional plasticity in disease, whereas large cholangiocytes are engaged in hormone-regulated bile secretion.
Cholangiocytes play an important role in the formation and transportation of bile through transmembrane molecules expressed on the basolateral or aplical membrane. These cells interact with both resident and non-resident cells of the bile duct via fibrotic and inflammatory mediators, such as interleukin 6 (IL-6) and tumor necrosis factor α (TNF-α).
The tissue homeostasis in the biliary system is maintained via a cell-cycle phenomenon linked with cholangiocytes. The interaction between cholangiocytes and various modulators of apoptosis, senescence, and proliferation, such as platelet-derived growth factor, helps in tissue homeostasis.
Cholangiopathy: a biliary disorder
The concept of cholangiopathy has emerged from the abnormal functioning of cholangiocytes. A diseased or damaged cholangiocyte leads to fibrosis, inflammation, dysplasia, ductopenia, or malignant transformation of the bile ducts.3
Cholangiopathy has been classified into immune-mediated states, genetic, infectious, malignant, idiopathic, and secondary sclerosing cholangitis.4 Although each of these cholangiopathies has a unique clinical course, they share common processes that involve innate immune responses, proinflammatory signaling, tissue repair processes, and cholangiocyte proliferation and differentiation.
Cholangiocytes are activated via various insults that enhance the expression of proinflammatory cytokines and chemokines. These molecules function in autocrine and paracrine manners and induce cholangiocyte proliferation, senescence, fibrosis, apoptosis, and angiogenesis, and recruit innate and adaptive immune cells, endothelial cells, and mesenchymal cells. These events trigger ductal reactions, such as infiltration of leukocytes and lymphocytes, the proliferation of ductules, activation of liver progenitor cells, and an increase in matrix protein, and ultimately cause ductopenia, periportal fibrosis, and biliary cirrhosis.5
Host factors depend on epigenetics, genetic susceptibility, or posttranscriptional regulation for repair/resolution or chronic inflammation of the bile ducts. Cholangiopathies share two major features, i.e., an imbalance between cell proliferation and survival and a selective target of either small or large bile ducts. Primary biliary cirrhosis (PBC), primary sclerosing cholangitis (PSC), cystic fibrosis, biliary atresia, and polycystic liver disease are common cholangiopathies.6
Image Credit: Alina.Alina/Shutterstock.com
Some patients have been diagnosed with post-coronavirus disease 2019 (COVID-19) cholangiopathy. This condition has been referred to as COVID-19-associated secondary sclerosing cholangitis. This is a relatively new and under-recognized disease entity. Therefore, more research is required to establish protocols for early diagnosis and management of patients with post-COVID-19 cholangiopathy.7
Clinical breakthroughs against cholangiopathies
It is extremely important to understand the biology of cholangiocytes to uncover the mechanism of cholangiopathy, which will positively help formulate effective treatment for many of these diseases. The manner by which cholangiocyte responds to injury is regulated by bile acids in conditions of ductopenia and cholestasis.8 Put simply, bile acids exert cytoprotective effects.
Ursodeoxycholic acid (UDCA) has shown a significant effect in alleviating PBC condition but not PSC. UDCA normalizes serum markers of liver dysfunction and inhibits histologic disease progression. A recent study has documented that nor-ursodeoxycholic acid (nor-UDCA) and obeticholic acid (OCA) could be used to treat cholestatic liver diseases.9
OCA is a semisynthetic analog of the bile acid chenodeoxycholic acid that possesses a robust affinity for the farnesoid X receptor (FXR). This receptor is mainly expressed in the liver and gut, which regulates complex signaling pathways associated with the homeostasis of bile acids.
Nor-UDCA is a novel therapeutic candidate for cholangiopathies that exhibited significant efficacy against sclerosing cholangitis in Mdr2 knockout mice. The mode of action of this drug candidate is linked to increased hydrophilicity of bile acids, which leads to detoxification and elimination of bile acids.
In the late stage of PBC, estrogen receptor expression gets significantly reduced. Since cholangiocytes express estrogen receptors and exert cytoprotective effects, PBC is more frequent in women. The clinical manifestation of this disease occurs after menopause.10
Endogenous opioid peptides are synthesized and locally released by reactive cholangiocytes. This peptide causes pruritus in cholestatic patients. Administration of opiate antagonists has effectively reduced pruritus in these patients. Reactive cholangiocytes also locally release serotonin which suppresses biological response to injury. This condition is reversed by administration of sertraline.
In response to injury, cholangiocytes release vascular endothelial growth factor (VEGF) and insulin-like growth factor–1 (IGF–1) that stimulate cholangiocarcinoma cell growth. Antiangiogenic therapies could be effective in cholangiocarcinoma.11 Monoclonal antibodies could be an effective tool to treat cholangiopathies, including PBC. At present, clinical trials are being conducted to assess the effectiveness of ustekinumab, an anti-p40 monoclonal antibody, against PBC.12
References
- Almajid AN, Sugumar K. Physiology, Bile. StatPearls. 2022; https://www.ncbi.nlm.nih.gov/books/NBK542254/. Accessed January 5 2024.
- Loarca L. et al. Development and characterization of cholangioids from normal and diseased human cholangiocytes as an in vitro model to study primary sclerosing cholangitis. Lab. Investig. 2017; 97(11), 1385-1396.
- Tam KHP. et al. Cholangiopathies – Towards a molecular understanding.EBioMedicine. 2018; 35, 281-393.
- Lazaridis KN, LaRusso NF. The cholangiopathies. Mayo Clin Proc. 2015; 90:791–800. doi: 10.1016/j.mayocp.2015.03.017.
- Tietz PS, Chen XM, Gong AY. et al. Experimental models to study cholangiocyte biology. World J Gastroenterol. 2002; 8:1–4.
- Maroni, L. et al. Functional and Structural Features of Cholangiocytes in Health and Disease. Cell. Mol. Gastroenterol. Hepatol. 2016; 1(4), 368-380. https://doi.org/10.1016/j.jcmgh.2015.05.005
- Kim HY and Lee SS. Post-COVID-19 Cholangiopathy: Clinical and Radiologic Findings. KJR. 2023; 24(11), 1167-1171. https://doi.org/10.3348/kjr.2023.0832
- Yoo S, Lim W T, and Choi H S. Biology of Cholangiocytes: From Bench to Bedside. Gut and Liver. 2016; 10(5), 687-698. https://doi.org/10.5009/gnl16033
- Floreani A, Gabbia D, and Martin S D. Obeticholic Acid for Primary Biliary Cholangitis. Biomedicines. 2022; 10(10). https://doi.org/10.3390/biomedicines10102464
- Marzioni M, Ueno Y, Glaser S. et al. Cytoprotective effects of taurocholic acid feeding on the biliary tree after adrenergic denervation of the liver. Liver Int. 2007; 27:558–568. doi: 10.1111/j.1478-3231.2007.01443.x.
- Pellicciari R, Fiorucci S, Camaioni E. et al. 6alpha-ethyl-chenode-oxycholic acid (6-ECDCA), a potent and selective FXR agonist endowed with anticholestatic activity. J Med Chem. 2002; 45:3569–3572. doi: 10.1021/jm025529g.
- Loeff FC. et al. Clinical Impact of Antibodies against Ustekinumab in Psoriasis: An Observational, Cross-Sectional, Multicenter Study. JID. 2020; 140(11), 2129-2137. https://doi.org/10.1016/j.jid.2020.03.957
Further Reading
Last Updated: Jan 24, 2024