The primary aim of any vaccine is to stimulate immune functions and confer long-lasting protection. A paper recently uploaded to the preprint server bioRxiv* by Tong et al. (17th Feb 2021) describes a novel vaccine utilizing PIKA adjuvant and recombinant spike proteins against COVID-19 disease that promises the generation of potent neutralization antibodies, which potentially support immune responses for years.
What are recombinant vaccines?
Recombinant proteins are created in genetically engineered cells, modified to express the protein of interest. In this case, the group chose the full-length spike protein of the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), as it contains many regions able to induce virus-specific T-cells. The spike protein was modified by two proline mutations to keep it in its pre-fusion form, a similar technique to that adopted by several other groups developing vaccines. PIKA was also incorporated into the final vaccine, and mice were administered weekly over a three-week period with 5 μg of protein and 50 μg of PIKA. The group found that antibodies specific to the spike protein were detected at a titer of more than 50,000 at the peak of day 21, with an average of 1,000, purportedly higher than achieved by the three approved vaccines (Moderna, Oxford-AstraZeneca, and Pfizer-BioNTech).
Polyinosinic: polycytidylic acid (poly I:C) is a synthetically produced mismatched dsRNA strand that interacts with toll-like receptors on the surface of immune cells to act as an antiviral agent. It is usually referred to as PIKA when used as a vaccine adjuvant in a stabilized form and has been demonstrated to provide quantitative and qualitative improvements to immune responses when coadministered with an unadjuvanted vaccine.
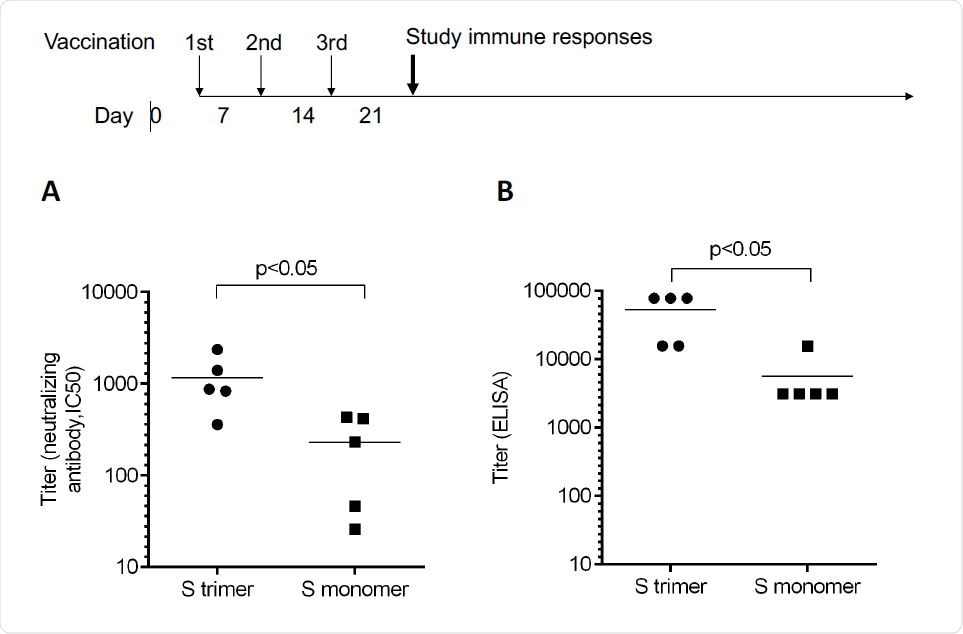
Antibody titers measured by neutralizing assays and ELISA. Mice were immunized by Spike trimer or monomer proteins containing PIKA adjuvant by a 0-7-14 day schedule. A. The neutralizing titer as measured by pseudo-virus with monoclonal antibodies 40592-MM57 and 40591-MM43 as control (with IC50 at 1 μg/ml). B. Antibody titer as measured by ELISA using plate-bound trimeric Spike protein.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
What are the advantages of recombinant vaccines?
A protein/peptide array was performed to test the method of binding, and showed that the produced antibodies that bound to the engineered spike protein did so similarly to the natural spike protein, mainly around the receptor-binding domain. This demonstrates that the recombinant protein is able to stimulate the immune system similar to the real virus, conferring effective and long-lasting immunity.
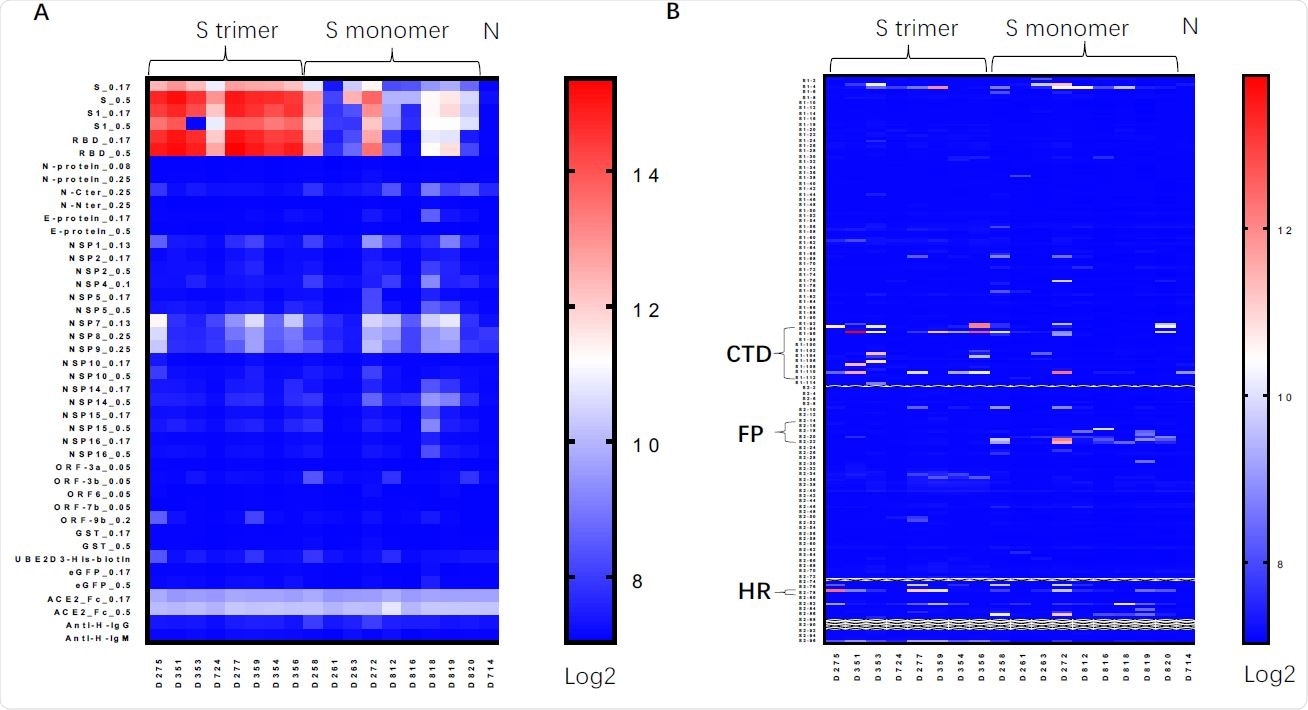
Protein/peptide array of serum antibodies induced by Spike protein vaccines. A. Protein array assay for sera from mice immunized by Spike trimer, Spike monomer, Non-immunized mice, using Spike (S_0.17 and S_0.5 means proteins were printed at 0.17 or 0.5 mg/ml); S1 subunit of Spike, RBD and other viral proteins. B. Linear peptide array using linear peptides of Spike proteins. CTD, C-terminal domain right after RBD (Peptides S1-93-S1-113); FP, fusion peptide (Peptides S2-14-S2-23); HR, heptad regions (Peptides S2-78).
The group used Chinese hamster ovary (CHO) cells to express the recombinant protein. CHO cells are popularly used in the production of recombinant proteins as they are easy to culture on a large scale and produce prodigious quantities of the desired protein, up to 10 grams per liter of culture. Many decades of working with these cells have allowed detailed genetic engineering that produces human biocompatible molecules.
Both recombinant protein and mRNA vaccines require the use of adjuvants, such as PIKA, in this case, to stimulate a robust immune response and confer long-lasting immunity. However, recombinant protein vaccines do not need to be stored at -80⁰C, as with the two most popular COVID-19 mRNA vaccines by Moderna and Pfizer-BioNTech. Other vaccines based on recombinant proteins have seen success with longer-lasting immunity than conferred by mRNA vaccines, including hepatitis B and HPV vaccines. Comparatively, manufacturing is continuous, more readily scalable, and cheaper.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Journal references:
- Preliminary scientific report.
Potent neutralization antibodies induced by a recombinant trimeric Spike protein vaccine candidate containing PIKA adjuvant for COVID-19, Jiao Tong, Chenxi Zhu, Hanyu Lai, Chunchao Feng, Dapeng Zhou bioRxiv 2021.02.17.431647; doi: https://doi.org/10.1101/2021.02.17.431647, https://www.biorxiv.org/content/10.1101/2021.02.17.431647v1
- Peer reviewed and published scientific report.
Tong, Jiao, Chenxi Zhu, Hanyu Lai, Chunchao Feng, and Dapeng Zhou. 2021. “Potent Neutralization Antibodies Induced by a Recombinant Trimeric Spike Protein Vaccine Candidate Containing PIKA Adjuvant for COVID-19.” Vaccines 9 (3): 296. https://doi.org/10.3390/vaccines9030296. https://www.mdpi.com/2076-393X/9/3/296.