In a recent study published in the journal PNAS, a group of researchers developed an intranasal measles-mumps-SARS-CoV-2 spike (S) protein (MMS) vaccine that offers broad, durable protection against major Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) variants, leveraging the proven safety of the measles-mumps-rubella (MMR) vaccine platform.
 Study: A next-generation intranasal trivalent MMS vaccine induces durable and broad protection against SARS-CoV-2 variants of concern. Image Credit: TopMicrobialStock / Shutterstock
Study: A next-generation intranasal trivalent MMS vaccine induces durable and broad protection against SARS-CoV-2 variants of concern. Image Credit: TopMicrobialStock / Shutterstock
Background
The coronavirus disease 2019 (COVID-19) pandemic, triggered by SARS-CoV-2, has led to over 6.96 million deaths and more than 771 million infections globally as of October 2023. Although several vaccines using the prefusion spike (S) protein as an immunogen have been developed, they have limitations like reduced effectiveness against evolving SARS-CoV-2 variants, short-lived protection, and lack of mucosal immunity. The emergence of various variants, especially the Omicron with numerous mutations, poses challenges to current vaccines' effectiveness. Given this, there is a pressing need for more effective vaccines. Given the persistent emergence of SARS-CoV-2 variants and the limitations of current vaccines, further research is needed for next-generation intranasal vaccines that provide broader mucosal immunity and are adaptable to evolving strains.
About the study
The research delved into the comprehensive analysis and development of modified measles and mumps viruses (recombinant Measles Virus (rMeV), recombinant Mumps Virus (rMuV-JL1), and rMuV-JL2) that express the SARS-CoV-2 six prolines (preS-6P) proteins. Procedures for growth curves, virus preparation, purification, and various assays were implemented. These assays include Measles Virus (MeV), Mumps Virus (MuV), and SARS-CoV-2 plaque tests, ribonucleic acid (RNA) extraction, Reverse Transcription Polymerase Chain Reaction (RT-PCR), and Western blotting, among others. The Ohio State University's Institutional Laboratory Animal Care and Use Committee approved the animal studies, referencing protocol numbers 2009A1060-R3 and 2020A00000053. Animal testing involved immunizing and challenging IFNAR1−/− mice and golden Syrian hamsters.
Further steps in the study detailed protein purification, T cell assays, flow cytometry, and Enzyme-Linked Immunosorbent Assay (ELISA) tests to detect specific SARS-CoV-2 Immunoglobulin G (IgG) and Immunoglobulin A (IgA) antibodies. They also explored SARS-CoV-2's neutralizing ability, pseudotype neutralization, and the determination of SARS-CoV-2 concentration in animal tissues. Histological examinations were conducted, and all results underwent statistical analysis.
Study results
Researchers recently found that the preS stabilized with preS-6P induced a higher neutralizing antibody response than another variant. The preS-6P gene of the SARS-CoV-2 Delta variant was then integrated into a different genome. This altered strain differed in growth and cellular effects but maintained strong protein expression, similar to the Delta variant. A gene from the SARS-CoV-2 Omicron BA.1 was also integrated into a separate vaccine genome with similar observations.
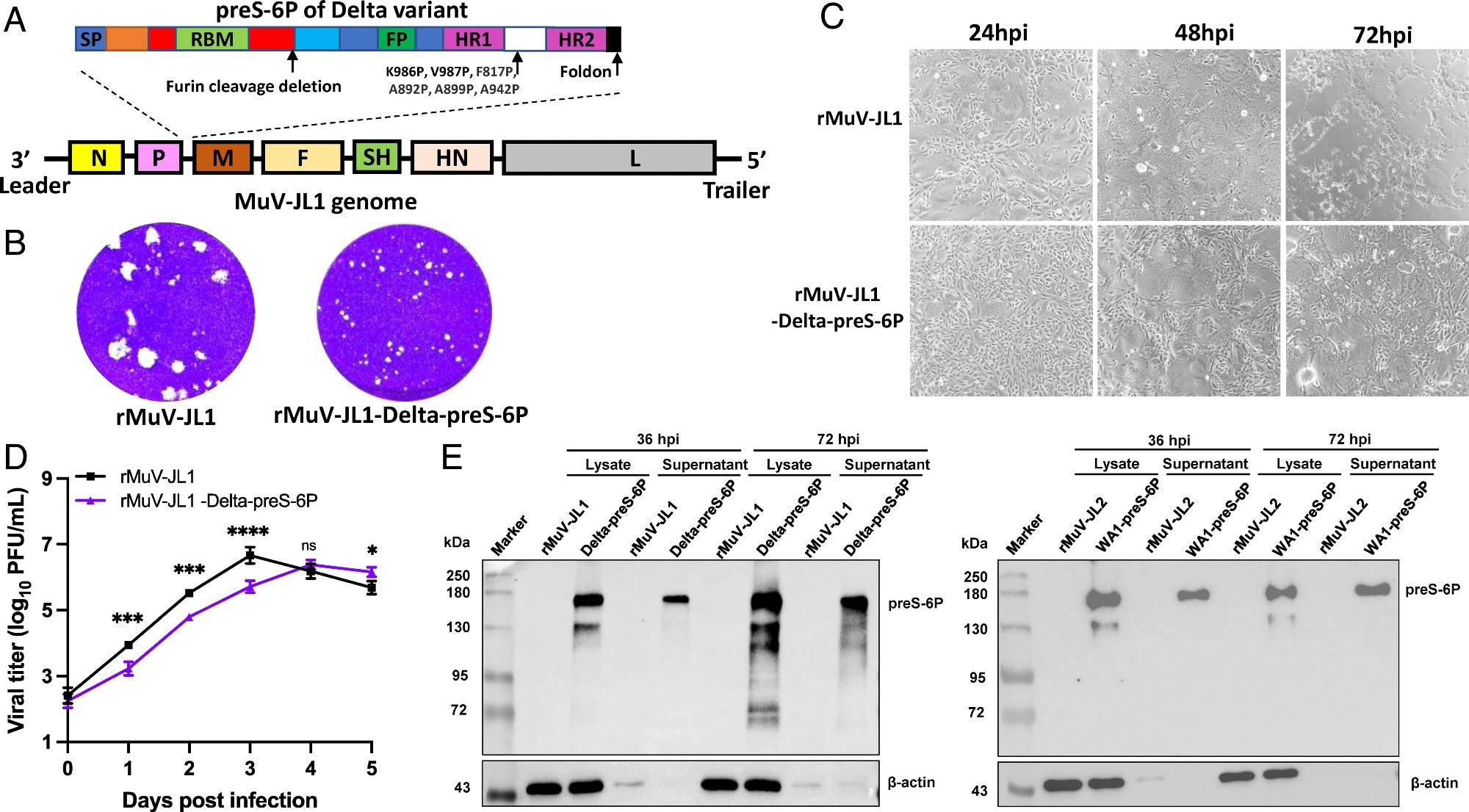 Recovery and characterization of rMuV-JL1-Delta-preS-6P expressing the six proline-stabilized prefusion spike of SARS-CoV-2 Delta variant. (A) Strategy for insertion of preS-6P of the Delta variant into the P and M gene junction in the MuV-JL1 genome. (B) The plaque morphology of rMuV-JL1 and rMuV-JL1-Delta-preS-6P in Vero CCL81 cells at day 5. (C) rMuV-JL1-Delta-preS-6P exhibits delayed syncytia formation in Vero CCL81 cells (MOI of 0.1). (D) Replication kinetics of recombinant viruses in Vero CCL81 cells at an MOI of 0.1. (E) Expression of preS-6P in rMuV-JL1-Delta-preS-6P (Left) or rMuV-JL2-WA1-preS-6P (Right)-infected Vero CCL81 cells. An MOI of 0.1 was used for infection, and 10 μL of cell lysate (from total 200 μL) and 10 μL (from total 1 mL) of supernatant were used for Western blot.
Recovery and characterization of rMuV-JL1-Delta-preS-6P expressing the six proline-stabilized prefusion spike of SARS-CoV-2 Delta variant. (A) Strategy for insertion of preS-6P of the Delta variant into the P and M gene junction in the MuV-JL1 genome. (B) The plaque morphology of rMuV-JL1 and rMuV-JL1-Delta-preS-6P in Vero CCL81 cells at day 5. (C) rMuV-JL1-Delta-preS-6P exhibits delayed syncytia formation in Vero CCL81 cells (MOI of 0.1). (D) Replication kinetics of recombinant viruses in Vero CCL81 cells at an MOI of 0.1. (E) Expression of preS-6P in rMuV-JL1-Delta-preS-6P (Left) or rMuV-JL2-WA1-preS-6P (Right)-infected Vero CCL81 cells. An MOI of 0.1 was used for infection, and 10 μL of cell lysate (from total 200 μL) and 10 μL (from total 1 mL) of supernatant were used for Western blot.
A trivalent vaccine was developed and tested against a single-strain vaccine in mice from these modified viruses. The trivalent vaccine led to a broader immune response, generating neutralizing antibodies against multiple SARS-CoV-2 variants. Both vaccines maintained antibody responses for four months and stimulated tissue-resident memory T cell responses in the lungs, critical for SARS-CoV-2 defense. However, the trivalent version showed a more comprehensive protective capacity.
Seven weeks post-immunization, T-cell responses were analyzed in the mice's splenocytes. The trivalent vaccine activated T-helper 1 (Th1) cells more than its monovalent counterpart and notably stimulated more Interleukin-4 (IL-4)-producing T helper cells. Both vaccines incited significant numbers of T helper cells producing IL-21 and IL-17. Still, the trivalent version demonstrated a superior systemic T-cell immune response.
In experiments using Golden Syrian Hamsters, the trivalent vaccine exhibited broader neutralizing activity against various SARS-CoV-2 variants. In contrast, the monovalent vaccine was more specific to the Omicron BA.1 variant. When exposed to SARS-CoV-2, the trivalent vaccine offered enhanced protection, especially against the WA1 and Delta strains.
Additionally, intranasal administration of the trivalent vaccine in separate tests provided complete SARS-CoV-2 protection and triggered a more potent serum IgG antibody response compared to subcutaneous administration. Intranasal delivery also led to a potent mucosal IgA antibody response, not seen in the subcutaneous group. This indicates that the delivery method can significantly impact immune responses, with intranasal immunization potentially offering superior protection against respiratory viruses like SARS-CoV-2.
Conclusions
To summarize, researchers developed an intranasal trivalent MMS vaccine that can combat measles, mumps, and various SARS-CoV-2 variants. The vaccine was observed to produce strong systemic and lung-specific immune responses, offering protection against multiple SARS-CoV-2 strains, including the Delta and Omicron BA.1 variants. This trivalent vaccine is a modified version of the MMR vaccine and showcases broad neutralizing capabilities against multiple virus strains, in contrast to monovalent vaccines, which are limited in their protective scope. Intranasal administration of the MMS vaccine induces strong immune responses both systemically and in the mucosal regions, potentially offering superior protection against SARS-CoV-2 variants. Importantly, the vaccine's design allows for easy modifications to address newly emerging variants. In essence, this next-generation COVID-19 vaccine candidate demonstrates broad and durable protection, highlighting its potential as a significant tool against SARS-CoV-2 variants.