Highly pathogenic avian influenza (HPAI) is a severe viral disease caused by subtypes (H5 and H7) of type A. Over the last century, these viruses have caused repeated endemic waves, spreading rapidly and causing substantial losses of avian fauna across a wide range of species, especially human-reared poultry.
Since 2003, however, HPAI A (H5N1) has been observed to breach kingdom barriers and jump from their avian hosts to mammals, resulting in two unpreceded panzootic events – 2003 to 2019 and the ongoing 2020 through 2023 period. These events have triggered alarm bells within the scientific community due to mutant H1N5 strains affecting endangered wildlife, the economic losses associated with their infecting livestock, and their potential for human transmission.
Limited reports on the ongoing panzootic period suggest that it is significantly more severe than the 2003 one. It is projected to be one of the worst panzootic events in recorded history across economic, geographic coverage, and animal morbidity and mortality scales. In the three years since the advent of the panzootic, the virus has spread across four continents and a record 26 countries, notably infecting minks, foxes, ferrets, seals, and domestic cats, all of which present the potential for human transmission.
Unfortunately, research on H1N5 and the ongoing panzootic remains scarce and restricted to ‘gray literature’ (non-peer-reviewed records and reports from government databases and websites). Understanding the evolution of the H5N1 virus and the mutations allowing emergent strains to far outdo their ancestral virus in infectivity and species spread would better equip policymakers and scientists alike with the information required to attenuate the ongoing panzootic before it explodes into a full-fledged pandemic.
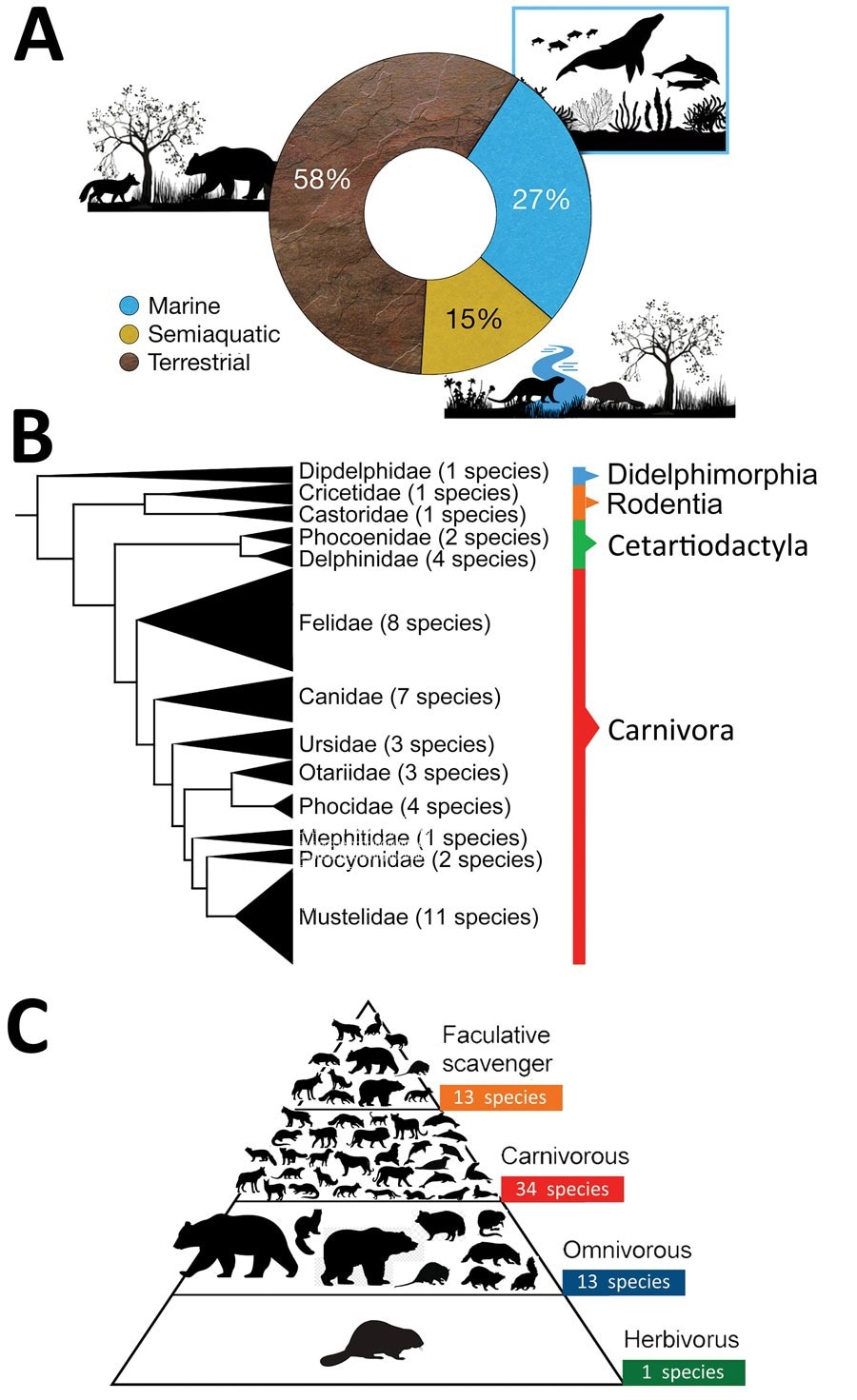
Characteristics of mammal species affected worldwide by highly pathogenic influenza virus A (H5N1) the current panzootic (2020–2023). A) Habitat of mammal species affected by H5N1. B) Phylogeny of mammal species affected (tree constructed using iTOL version 5 following Letunic and Bork, from DNA sequence data available in Upham et al.). C) Trophic level (facultative scavenger, carnivore, omnivore, or herbivore) of mammal species affected worldwide by H5N1. Some of the omnivorous and carnivorous mammals included in the pyramid (n = 13) also consume carrion; thus, they are also considered to be facultative scavengers and are incorporated in the upper part of the pyramid.
About the study
In the present review, researchers compiled and analyzed scientific literature on natural mammalian H5N1 infections (including humans) and compared findings from the current panzootic to those from previous H5N1 waves. The review focuses on the number and habitats of infected species, their phylogeny, infection sources, and necropsy findings. They further investigate the viral mutations that enable cross-species transmission and elucidate potential risks to biodiversity and human health.
Data was collected from the Scopus and Google Scholar online databases, with searches divided into two periods – 1996 to 2019 and 2020 to 2023. Studies based on serologic evidence were excluded from analyses due to uncertainty on the time of infection, which may bias diagnostic results. Additionally, the World Organisation for Animal Health, the United Kingdom’s Animal and Plant Health Agency, and the United States of America (US) Department of Agriculture’s Animal and Plant Health Service were queried for up-to-date information on the current panzootic.
Data from the World Health Organization (WHO) was collated for information on human infections. Finally, conservation statuses of infected species were derived from the International Union for Conservation of Nature (IUCN) Red List of Threatened Species, their diets and habitats from MammalBase, and H5N1 sequence data from Upham et al. (2019).
Study findings
The database literature review revealed 59 publications on mammalian H5N1 infections, 23 of which discussed previous H5N1 waves and 36 on the ongoing panzootic. Scientific interest in the ongoing panzootic is immediately evident – more mammalian infection data has been generated in the last three years than was generated in the preceding 23.
Alarmingly, while previous waves combined reported 10 infected countries spread across three continents (Asia, Europe, and Africa), the ongoing panzootic has already spread to 26 countries across Europe, South America, North America, and Asia. Limited testing and reporting from other nations suggests that these findings are underestimations.
“Our review suggests that H5N1 virus is expanding its geographic range to new continents such as North and South America. This fact is of concern because when an emerging pathogen reaches naive populations, the consequences for biodiversity can be catastrophic, especially for threatened species.”
Investigations into the number of species affected reveal that while previous panzootics cumulatively infected nine predominantly terrestrial and semi-aquatic species, the current panzootic has already been detected in more than 48 mammalian species, including 13 species of marine mammals. Peru, Chile, and Argentina have reported thousands of dead individuals of seals and similar mammals (e.g., the American sea lion [Otaria flavescens]), almost resulting in localized extinction events.
Costs to biodiversity are critical – thus far, avian influenza has affected four near-threatened, four endangered, three vulnerable, and one critically endangered species as compared to previous panzootics, which cumulatively infected two endangered and two vulnerable species.
Similarities to previous pandemics do exist – most affected mammals are carnivores (mainly apex- and mesopredators) and scavengers, corresponding to the most likely infection sources – close contact (including ingestion) with dead or dying birds or infected carrion.
…in the year 2004, a total of 147 tigers and 2 leopards housed in zoos in Thailand became infected and died after consuming infected chicken carcasses. In the current panzootic, the first case of H5N1infection in minks in Spain was probably caused by contact with infected birds (perhaps gulls).”
At least five publications have reported an alarming trend in viral adaptation – H5N1 strains with novel mutations that may allow for mammal-to-mammal transmission have been identified. If these strains spread, models suggest that a global pandemic may occur rapidly, causing unprecedented biodiversity and economic loss.
Finally, H5N1 has been found to have spilled over and infected at least 878 humans and resulted in 458 deaths (52% lethality) with close contact with livestock (especially poultry) deemed the main transmission route.
“So far, no evidence indicates human-to-human transmission, and the risk for a pandemic event still seems low. However, one of the most severe influenza viruses to have affected humans (i.e., Spanish influenza [1918–1919]) developed from an avian influenza virus that adapted to humans, a fact that should be considered when assessing the spillover risk. Governments must assume responsibility for protecting biodiversity and human health from diseases caused by human activities. If we hope to conserve biodiversity and protect human health, we must change the way we produce our food (poultry farming, in this specific case) and how we interact with and affect wildlife.”
Journal reference:
- Plaza PI, Gamarra-Toledo V, Rodríguez Euguí J, Lambertucci SA. Recent changes in patterns of mammal infection with highly pathogenic avian influenza A(H5N1) virus worldwide. Emerg Infect Dis. 2024 Mar, DOI – 10.3201/eid3003.23109, https://wwwnc.cdc.gov/eid/article/30/3/23-1098_article