With the COVID-19 pandemic, the world has suffered hundreds of thousands of deaths and tens of millions of cases. In order to contain this outbreak, new and already approved drugs are being tested. A new paper by researchers at Japan’s National Institute of Infectious Diseases and published on the preprint server bioRxiv* in August 2020 reports the in vitro suppression of coronavirus (CoV) replication by an inhaled corticosteroid (ICS) ciclesonide.
Steroids have been repurposed for the treatment of severe COVID-19 because the latter is associated with severe inflammatory phenomena. Thus, the researchers turned to this class of compounds to identify a specific inhibitor of the virus. Systemic steroids, however, are associated with clinical complications and increased mortality when used in cases of severe MERS or SARS pneumonia, due to their ability to suppress innate and adaptive immunity, resulting in increased viral replication.
Ciclesonide Inhibits MERS-CoV Growth
The current study is focused on a steroid compound that does not have this replication-promoting characteristic. An earlier paper from these researchers shows ciclesonide prevents viral replication, based on which findings, a retrospective cohort-based clinical trial is underway in Japan. There are several case reports on patients with COVID-19 pneumonia who were treated with ciclesonide and had a favorable outcome, without significant side effects.
The current paper provides a scientific rationale for these clinical trials. The researchers reviewed 92 steroidal molecules for their ability to inhibit MERS-CoV-induced cytopathic effects (CPE), and narrowed them down to four, including ciclesonide. These were found to produce over 95% survival of infected cells. All four have a monocyclic ring, either five- or six-membered, attached to the steroid core.
Secondly, they looked at eight steroid molecules concerning their ability to prevent the growth of MERS-CoV in Vero cells and their cytotoxic effects, using a broad range of concentrations. They observed that ciclesonide showed both low cytotoxicity and high growth-suppressive properties. Systemically used steroids like cortisone, prednisolone, and dexamethasone failed to suppress viral growth, as did the topical steroid fluticasone.
The researchers further found that ciclesonide operates after viral entry occurs. They found that it suppressed the replication of MHV-2, MERS-CoV, SARS-CoV, HCoV-229E, and SARS-CoV-2. They passaged it repeatedly through the cells 11 times in the presence of 40 μM ciclesonide vs. 40 μM mometasone. This led to the identification of mutant viruses that were resistant to ciclesonide but not to mometasone. The single amino acid substitution was recapitulated in a recombinant virus generated from the parental strain and showed resistance to the drug.
Antiviral Effects on SARS-CoV-2
At this point, the researchers shifted focus to SARS-CoV-2. They found that Vero E6 cells expressing TMPRSS2, and human bronchial epithelial Calu-3 cells, are very susceptible to the virus. They then prepared a lung-like air-liquid interface (ALI) for differential primary human bronchial-tracheal epithelial (HBTE) cells. They found that SARS-CoV-2 replicated rapidly within these cells, with the viral RNA load increasing 2,000 times by day 3 from infection.
Ciclesonide suppressed this rise at low concentrations, with the EC90 being 0.55 μM. They also found that in comparison to nelfinavir and lopinavir, which strongly suppress viral replication, this drug also has comparable efficacy, unlike fluticasone and dexamethasone.
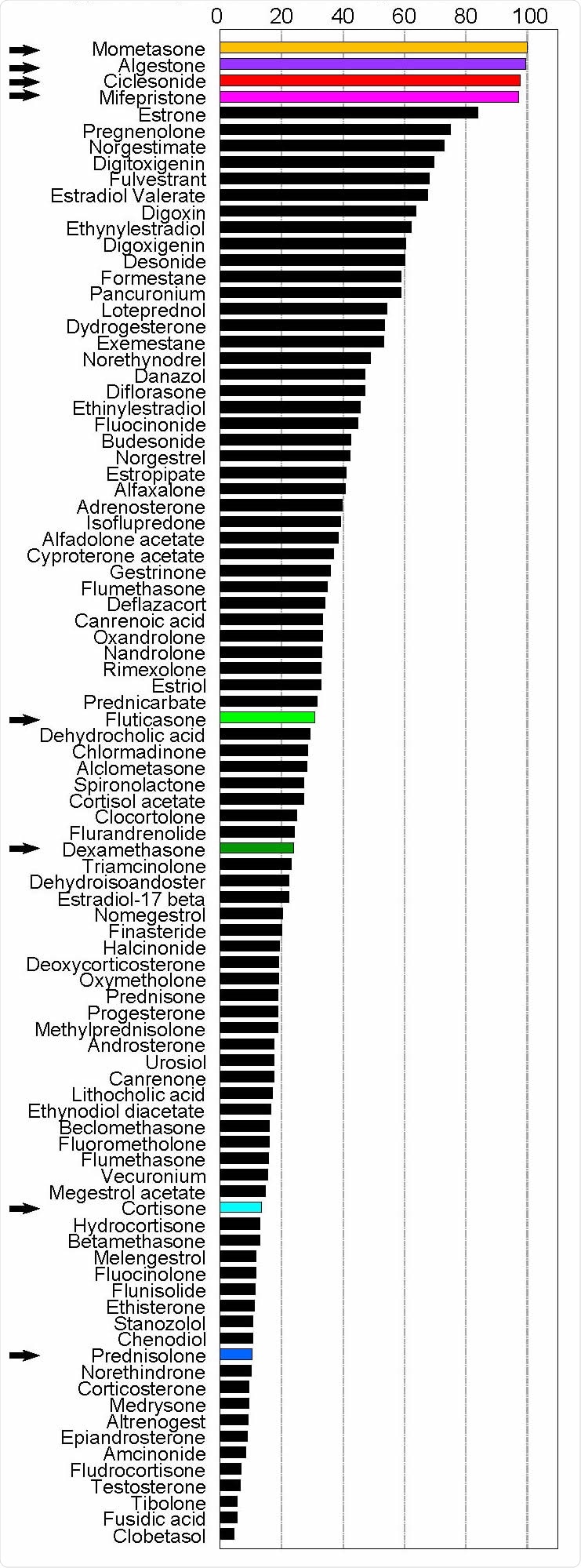
Steroid compounds reduce death rate of MERS-CoV infected cells. Vero cells seeded in 96- well microplates were infected with 100 TCID50 MERS-CoV in the presence of steroid compounds (10 μM). Cytopathic effects were observed at 72 h post-infection. Surviving cells were stained with crystal violet and photographed and quantified using ImageJ software. Data are presented as the average of two independent wells. Arrows indicate the steroid compounds assessed further in this study.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Ciclesonide Targets
The researchers then attempted to identify the molecular target of ciclesonide in its inhibition of viral replication. They used 43 viral isolates from infected patients to generate mutants that were resistant to the virus. They found that after passaging them through the Vero E6 cells eight times, in the presence of 40 μM ciclesonide, they had 15 single-mutation isolates. Whereas the amount of viral RNA decreased by a factor of 1,000 in the presence of ciclesonide and the parental virus. Though this decline was seen with the escape mutants also, the amount of viral RNA was 6- to 50-fold higher than with the parental virus. Thus, the parental mutants showed only a partial escape from ciclesonide blockage of replication.
The mutations in these escape mutants were found at four positions, one in nsp4 and three in nsp3. These proteins participate in forming double-membrane vesicles (DMV), to anchor the coronavirus replication-transcription complex inside the cells, and are inhibited by ciclesonide in a dose-dependent manner.
How It Works
A ciclesonide escape mutant of MERS-CoV showed an amino acid substitution at a site within nsp15, which is an RNA endonuclease responsible for its replication. An in silico study indicated that ciclesonide might act directly on this site to inhibit replication. In SARS-CoV-2, however, mutations were found in the C-terminal cytosolic domain of nsp3 or a large loop domain of nsp4. The former contains a protease which contributes to the central scaffolding protein of the replication transcription complex.
Implications
Ciclesonide is a safe drug and has been used in infants at high doses. This combination of antiviral potency with an excellent safety profile makes this a promising broad-spectrum antiviral activity against multiple CoVs. It has no significant systemic presence following inhalation and has weaker immunosuppressive effects than the potent systemic steroids cortisone and prednisolone.
This study indicates the potential for this drug to inhibit viral replication and the resulting inflammation in the lungs. Ciclesonide can suppress replication of multiple CoVs, and may, therefore, act as a broad-spectrum antiviral against this family of viruses, including the treatment of common colds, MERS, and COVID-19. The inhibitory concentration of ciclesonide required for suppression of replication in SARS-CoV-2 in HBTE cells is much lower than that required in VeroE6/TMPRSS2 or Calu-3 cells, perhaps because this is a prodrug that is metabolized in lung tissue (represented by HBTE cells) to its active form.
The study also shows how rapidly escape mutants emerge in patients on treatment with ciclesonide, but also the persistent suppression of replication by the drug by over 90% in these mutant strains as well. These novel mutants have not been identified in the GISAID and NCBI databases.
Though ciclesonide specifically suppresses the replication of CoVs, there is uncertainty about whether the drug inhibits DMV formation or RNA replication. Further research may uncover the exact mechanism of ciclesonide’s antiviral effects.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources