Increasing our understanding of cellular processes requires information about the types of biomolecules involved, their locations, and their interactions. This requires the molecules to be labeled without affecting physiological processes (bioorthogonality). This works when the markers are very quickly and selectively coupled using small molecules and “click chemistry”. In the journal Angewandte Chemie, a team of researchers has now introduced a novel type of click reaction that is also suitable for living cells and organisms.
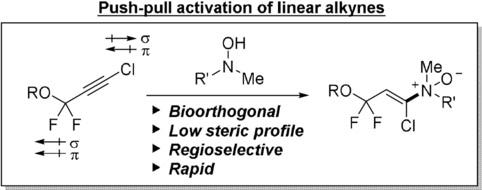
Image Credit: Angewandte Chemie
As an example, labeling biomolecules allows for the localization and characterization of tumors when an antibody that binds to specific molecules in the tumor cells is injected. A dye is then also injected. The antibodies and the dye are both equipped with small molecular groups that have almost no influence on cellular processes. When they encounter their counterpart, they bind immediately and specifically to each other with no side reactions—as easily as the two parts clicking together. This is where the term click chemistry comes from. The dye only remains attached to tumor cells, making them detectable.
The most well-known click chemistry reaction is the azide–alkyne reaction. An azide group reacts with an alkyne group to form a five-membered ring. However, this reaction requires a toxic copper catalyst, making it unsuitable for living systems. An alternative is the use of cyclic alkynes, in which the triple bond is under so much strain that the reaction works without a catalyst. Yet, the cycle can be unsuitable for some applications.
A team headed by Justin Kim at the Dana–Farber Cancer Institute and Harvard Medical School (Boston, USA) has now developed an alternative click reaction with linear, terminal alkynes, which works rapidly and is catalyst-free under complex physiological conditions. After a precise analysis of the electronic interactions in alkynes and tests with a variety of substituents, the team found that certain alkynes with halogens on both sides of the triple bond are reactive enough. The trick was to balance the different influences of the individual substituents so that the alkynes were sufficiently activated (push–pull activation) to react without a catalyst while remaining safe from attack by cellular components. For the other half of the click unit the team chose to use N,N-dialkylhydroxylamines (organic compounds containing both nitrogen and oxygen) instead of the conventional azides. The resulting reaction products (enamine-N-oxides) are biocompatible.
These new click reactions (retro-Cope eliminations) are very fast. The products are formed regioselectively, and the components are sufficiently stable and can easily be introduced to biomolecules. This broadens the spectrum of bioorthogonal coupling reactions for cellular labeling in living systems.
Source:
Journal reference:
Kang, D., et al. (2021) Bioorthogonal Hydroamination of Push–Pull-Activated Linear Alkynes. Angewandte Chemie. doi.org/10.1002/anie.202104863.