The worldwide spread of severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) has caused the global coronavirus disease 2019 (COVID-19) pandemic. To date, this pandemic has infected over 207 million individuals and claimed more than 4.36 million lives. The novel virus has a spike glycoprotein on its outer envelope, mediating the interaction between the virus and the human receptor, namely, angiotensin-converting enzyme 2 (ACE2). Subsequently, the virus gains entry into the host cell.
Toll-like receptor 4 (TLR4) is an important pattern recognition receptor (PRR) associated with the innate immune system. TLR4 is expressed on tissue-resident cells as part of the host’s intrinsic defense against harmful pathogens. The primary function of this receptor protein is to identify specific pathogenic (bacterial, viral, fungal, and parasite) components, known as pathogen-associated-molecular-patterns (PAMPs), and trigger cellular and systematic immune responses.
Activation of TLR4 is associated with the secretion of chemokines and pro-inflammatory cytokines, which cause inflammation. In addition, activation of TLR4 also results in the secretion of type 1 interferons (e.g., b-interferon), whose primary role is to alert neighboring cells about viral infection and inhibit viral replication.
TLR4 is present in both cell surfaces and endosomes. TLR4 is the only TLR that has canonical and alternative downstream signaling pathways: (a) a canonical MyD88-dependent pathway involves the release of pro-inflammatory cytokines and chemokines and (b) the TRIF/TRAM-dependent endosomal pathway results in the secretion of type I interferons and some anti-inflammatory cytokines. Both these pathways are critical because they help regulate the immune response.
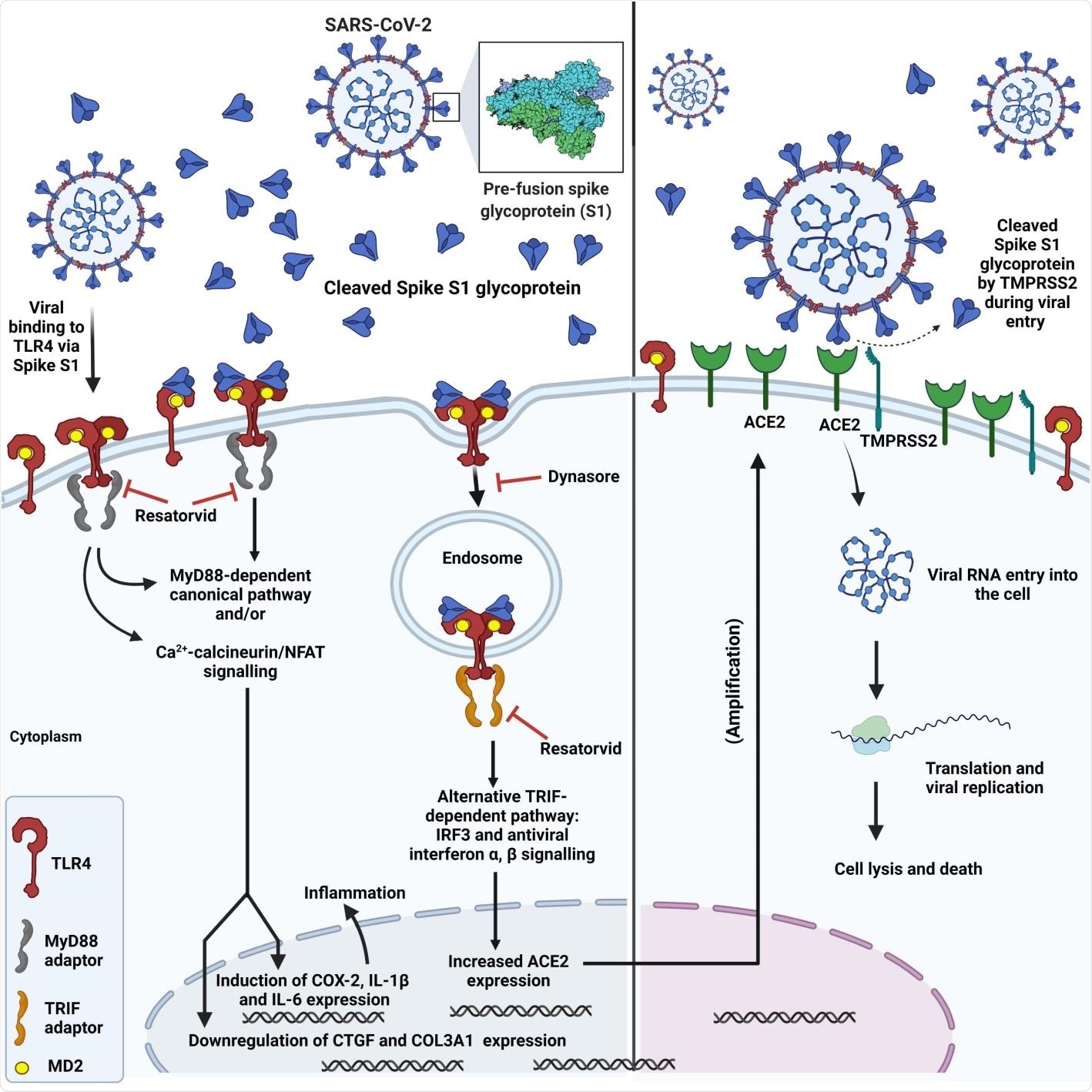
Schematic showing SARS-CoV-2 spike interaction with TLR4, subsequent TLR4 activation via canonical and endosomal pathways and induction of inflammation, upregulation of ACE2 expression and increased viral entry.
TLR4 and SARS-CoV-2 Infection
Prior studies have revealed that expression of ACE2 in the lung epithelial cells is low. However, SARS-CoV-2 is a respiratory disease, which causes hyperinflammation, known as cytokine storm, in severely infected individuals.
Such a contradiction has raised a question regarding the pathophysiology of SARS-CoV-2. A previous piece of research associated with COVID-19 infection revealed SARS-CoV-2 gains entry into the host cell by activating the TLR4 pathway at the early stage of infection.
The spike glycoprotein binds and activates TLR4, a transmembrane protein and a member of the toll-like receptor family, and increases ACE2 expression. This further assists in SARS-CoV-2 infection. Additionally, myocarditis and hyperinflammation, found in severely infected COVID-19 patients, may occur due to over-activation of TLR4 by the spike glycoprotein, which results in excessive release of pro-inflammatory cytokines downstream of TLR4.
A New Study
A group of researchers re-evaluated their previously proposed hypothesis, which stated that the TLR4 activated by the spike glycoprotein S1 domain increases ACE2 expression and, thereby, aids in COVID-19 infection.
A new study, published on the bioRxiv* preprint server, conducted in vitro studies using rat and human cells to test the original hypothesis. In this study, researchers revealed that the spike S1 domain of SARS-CoV-2 is a TLR4 agonist in rat and human cells, which is responsible for stimulating a pro-inflammatory M1 macrophage phenotype in human THP-1 monocyte-derived macrophages.
Main Findings
The SARS-CoV-2 transmembrane spike glycoprotein contains two subunits, namely, S1 and S2. In this study, the externally protruding S1 subunit was considered because, based on in silico molecular simulations, it contains the putative binding site to TLR4 and the Receptor Binding Domain (RBD), which binds to ACE2.
Previous studies have revealed that the S1 domain initially binds to ACE2 via the RBD. In contrast, the S2 domain mediates the fusion of the virus and host cell membranes, thereby leading to the entry of the virus into the host cell.
The current study hypothesizes that S1 activates TLR4 in neighboring cells during the initial infection. It has also highlighted the significance of the process of spike S1 domain being externalized from the cell.
This study has shown that spike S1 binds and activates TLR4 to increase ACE2 expression and triggers the release of pro-inflammatory cytokines, using adult rat cardiac tissue-resident macrophage-derived cells (cTMFs). cTMFs express TLR4 and co-express macrophage and myofibroblast markers.
Researchers have also used human cells expressing TLR4 (hTLR4-HA HEK 293 cells) and human monocyte-derived macrophages that express TLR4 to confirm their hypothesis.
The authors of the current study reported that the SARS-CoV-2 spike S1 subunit not only binds and activates TLR4, but also upregulates ACE2 expression in rat cTMFs.
This finding was derived using various techniques such as RT-qPCR, immunoblotting, confocal immunofluorescence microscopy, and proximity ligation assay.
Additionally, spike S1 caused downstream effects, analogous to bacterial lipopolysaccharide (LPS) on the expression of some inflammatory and fibrotic markers, e.g., cyclo-oxygenase-2 (COX-2), connective tissue growth factor (CTGF), and the collagen 3a1 isoform (COL3A1).
Researchers further reported that the effect of the spike S1 domain was inhibited by the selective TLR4 signaling inhibitor CLI-095 (Resatorvid® or TAK-242) which confirms that S1 is a TLR4 agonist and a viral PAMP for TLR4.
The binding of spike S1 to TLR4 in rat cTMFs and human TLR4 expressing cells was determined using proximity ligation assays. Further, inhibition of the expression of ACE2 by the dynamin inhibitor Dynasore® indicated that the endosomal/b-interferon pathway facilitates this expression.
Confocal immunofluorescence microscopy helped determine 1:1 stoichiometric spike S1 co-localization with TLR4 in rat and human cells. Researchers have also reported that spike S1/IFN-γ treatment of THP-1-derived macrophages triggers the pro-inflammatory M1 polarization, which was indicated by an increase in IL-1β and IL-6 mRNA.
Significantly, this work presents evidence for the further development of TLR4 antagonist drugs to improve the treatment of COVID-19 by reducing viral entry and inflammation.

 *Important notice: bioRxiv publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.
*Important notice: bioRxiv publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.