In a recent study published in the journal Nature, a group of researchers analyzed early cellular response dynamics to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in seronegative individuals using single-cell multi-omics profiling, identifying key cellular states and immune responses associated with different infection outcomes.
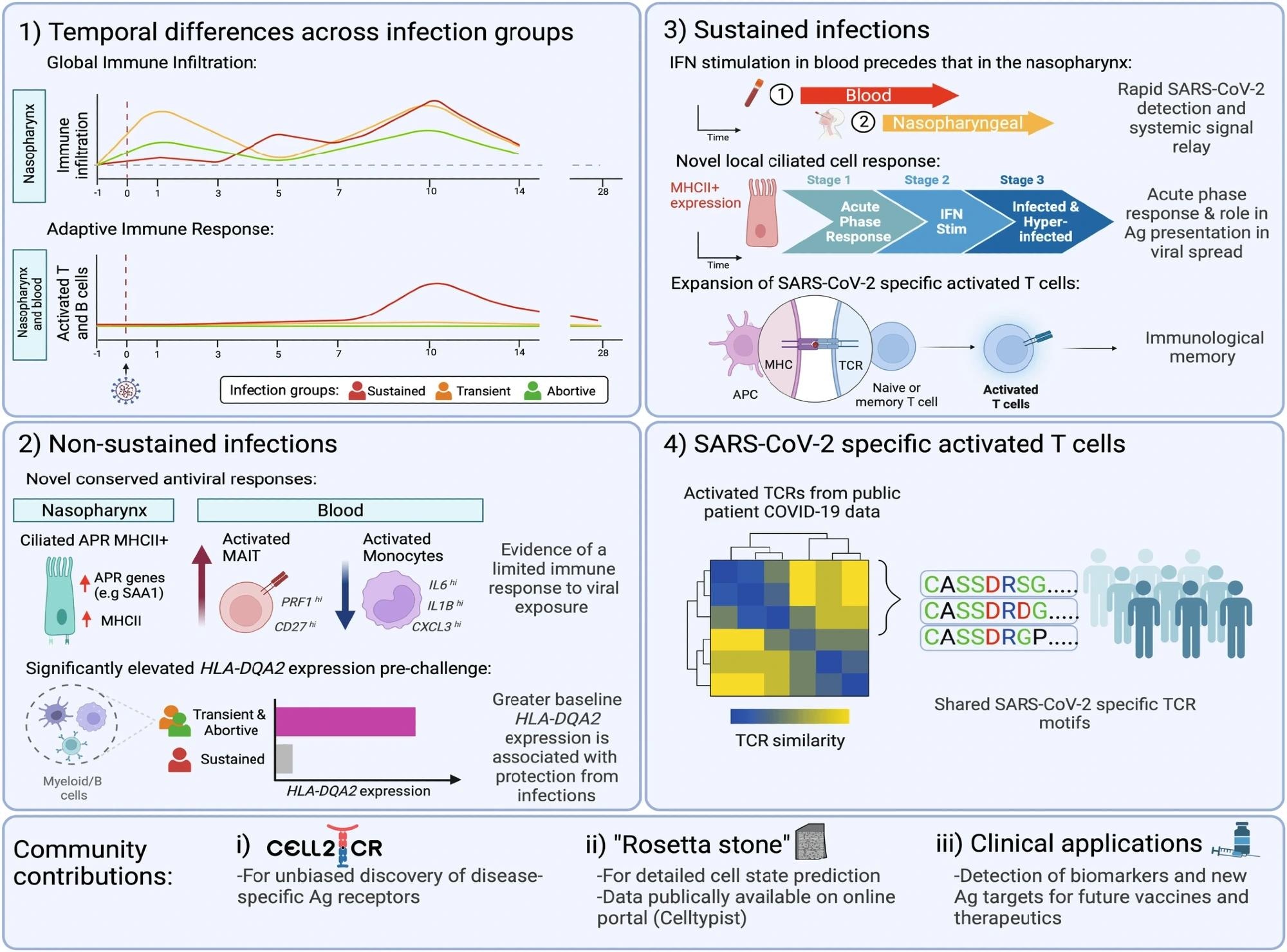 Summary figure highlighting the key finding from the study. These includes; 1) distinct temporal differences in the cellular dynamics observed between the different infection groups; 2) several novel conserved antiviral responses and higher baseline expression of HLA-DQA2 in participants who were exposed to the virus but who did not go on to develop a sustained infection; 3) novel characteristics of sustained infection, with a rapid relay observed in the blood compared to the site of inoculation, a dynamic local ciliated response occurring early on during infections (pre-symptoms) and a temporally restricted, distinct, SARS-CoV-2 specific activated T cell population which leads to immunological memory; and 4) the identification of public motifs in SARS-CoV-2 specific activated T cells. In addition, our work provides community tools for inference of specific TCR motifs (Cell2TCR) in activated T cells, a detailed publicly available reference database underpinning the detection of future biomarkers and antigen (Ag) targets for therapeutic applications. Schematic created with BioRender.com. Study: Human SARS-CoV-2 challenge uncovers local and systemic response dynamics.
Summary figure highlighting the key finding from the study. These includes; 1) distinct temporal differences in the cellular dynamics observed between the different infection groups; 2) several novel conserved antiviral responses and higher baseline expression of HLA-DQA2 in participants who were exposed to the virus but who did not go on to develop a sustained infection; 3) novel characteristics of sustained infection, with a rapid relay observed in the blood compared to the site of inoculation, a dynamic local ciliated response occurring early on during infections (pre-symptoms) and a temporally restricted, distinct, SARS-CoV-2 specific activated T cell population which leads to immunological memory; and 4) the identification of public motifs in SARS-CoV-2 specific activated T cells. In addition, our work provides community tools for inference of specific TCR motifs (Cell2TCR) in activated T cells, a detailed publicly available reference database underpinning the detection of future biomarkers and antigen (Ag) targets for therapeutic applications. Schematic created with BioRender.com. Study: Human SARS-CoV-2 challenge uncovers local and systemic response dynamics.
Background
Coronavirus 2019 (COVID-19), caused by SARS-CoV-2, is a potentially fatal disease that has become a severe global health emergency. Severe outcomes are linked to disrupted antiviral and immune responses, including impaired type I interferon responses and altered T lymphocyte (T) and B lymphocyte (B) cell dynamics. Accurate detection of immune responses is challenging due to heterogeneous factors such as viral dose, strain, and clinical features like comorbidities. Understanding the dynamics of SARS-CoV-2 infection, particularly early phases of exposure, is crucial. Studies often miss capturing these early events in natural infections, making pinpointing antigen-responding T cell activation and expansion difficult. Further research is needed to accurately delineate early immune response dynamics to SARS-CoV-2 to better understand and mitigate severe COVID-19 outcomes.
About the study
Sixteen healthy adults aged 18-30 years, seronegative for SARS-CoV-2, participated in a human SARS-CoV-2 challenge study for Single-cell RNA sequencing (scRNA-seq) sample processing and analysis. This study, conducted by a government task force, Imperial College London, Royal Free London NHS Foundation Trust, University College London, and hVIVO, took place from June to August 2021. Additionally, 20 healthy adults from earlier cohorts had blood and nasal samples processed for bulk Ribonucleic Acid (RNA)-seq, with 10 receiving pre-emptive remdesivir. Volunteers were screened for anti-SARS-CoV-2 antibodies and excluded if positive. The study followed ethical guidelines, and informed consent was obtained from all volunteers.
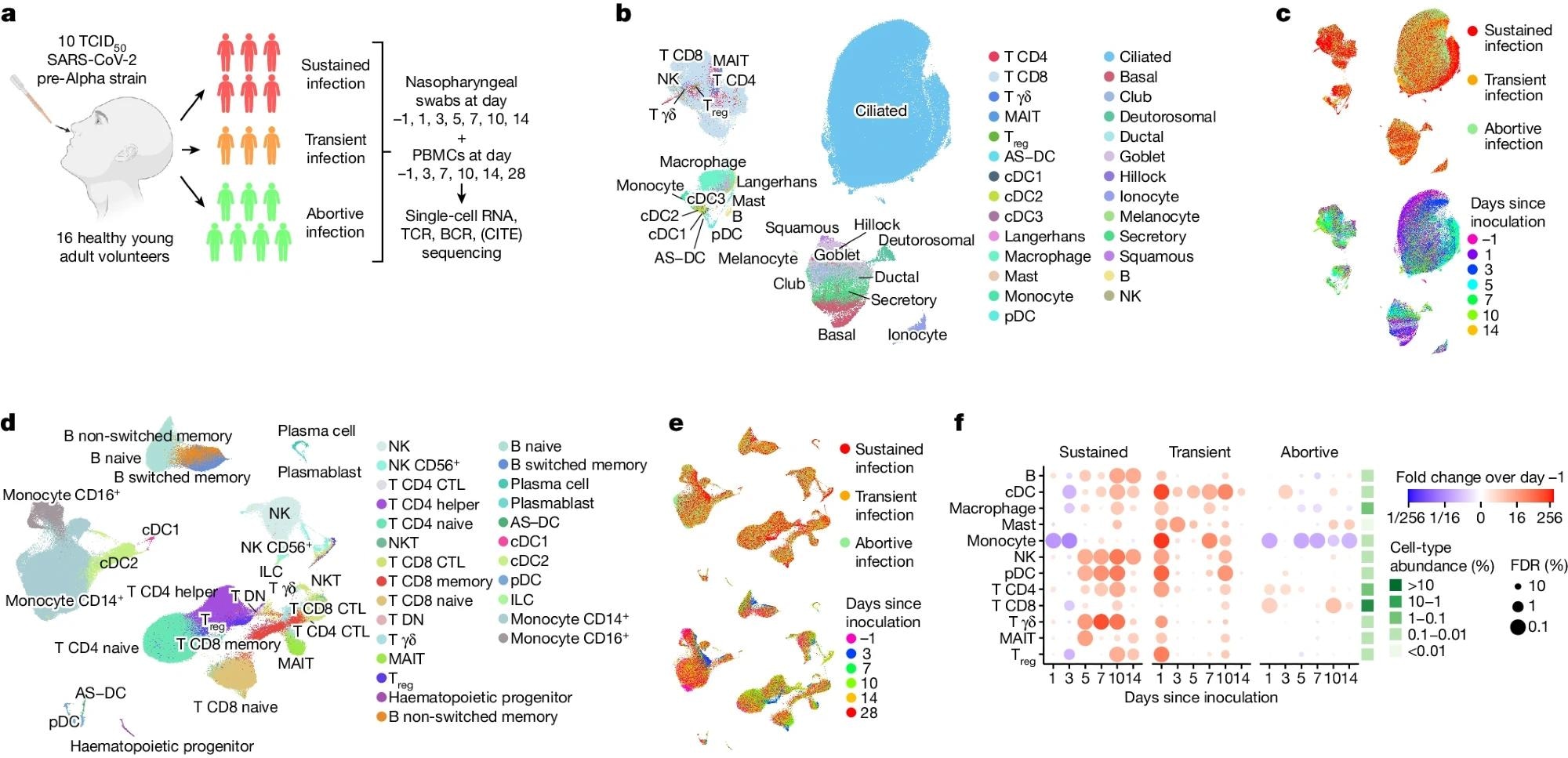 a, Illustration of the study design and cohort composition. b,c, Uniform manifold approximation and projection (UMAP) plots of all nasopharyngeal cells (n = 234,182), colour coded by their broad cell-type annotation (b), by the infection group (c, top) and by days since inoculation (c, bottom). Only cells from sustained infection cases are shown in c, bottom. Treg, regulatory T cell; AS–DC, AXL+SIGLEC6+ dendritic cell. d,e, UMAP plots as in b and c, but showing all PBMCs (n = 371,892). CTL, cytotoxic T lymphocyte; DN, double negative. f, Fold changes in abundance of nasopharynx-resident broad immune cell-type categories. Immune cell abundance was scaled to the total amount of detected epithelial cells in every sample before calculating the fold changes over days since inoculation compared with pre-infection (day –1) by fitting a GLMM on scaled abundance. Fitted fold changes are colour coded and we used the local true sign rate and Benjamini–Hochberg procedure to calculate false-discovery rates (FDRs), which are shown as the size of each dot. The mean cell-type proportions over all cells and samples are shown in the green heatmap to the right of the dot plot to aid the interpretation of changes in cell-type abundance. Illustration in a was created using BioRender (https://www.biorender.com).
a, Illustration of the study design and cohort composition. b,c, Uniform manifold approximation and projection (UMAP) plots of all nasopharyngeal cells (n = 234,182), colour coded by their broad cell-type annotation (b), by the infection group (c, top) and by days since inoculation (c, bottom). Only cells from sustained infection cases are shown in c, bottom. Treg, regulatory T cell; AS–DC, AXL+SIGLEC6+ dendritic cell. d,e, UMAP plots as in b and c, but showing all PBMCs (n = 371,892). CTL, cytotoxic T lymphocyte; DN, double negative. f, Fold changes in abundance of nasopharynx-resident broad immune cell-type categories. Immune cell abundance was scaled to the total amount of detected epithelial cells in every sample before calculating the fold changes over days since inoculation compared with pre-infection (day –1) by fitting a GLMM on scaled abundance. Fitted fold changes are colour coded and we used the local true sign rate and Benjamini–Hochberg procedure to calculate false-discovery rates (FDRs), which are shown as the size of each dot. The mean cell-type proportions over all cells and samples are shown in the green heatmap to the right of the dot plot to aid the interpretation of changes in cell-type abundance. Illustration in a was created using BioRender (https://www.biorender.com).
Participant 11, who had low pre-inoculation antibody levels, was classified as having an abortive infection, which did not alter the study's conclusions. Participants were followed for one year post-inoculation, with no long-COVID symptoms reported at the final time point. Physiological observations were normal. After discharge, two participants reported receiving a vaccine or a community infection before their day 28 follow-up. Enzyme-Linked ImmunoSpot (ELISpot) tests revealed immune responses in subsequent samples. Participants were intranasally inoculated with a wild-type pre-Alpha SARS-CoV-2 virus, and nasal and throat samples were collected to evaluate viral kinetics. Nasopharyngeal swabs and Peripheral Blood Mononuclear Cells (PBMCs) were processed for single-cell sequencing to analyze immune responses.
Study results
In the present study, 16 seronegative young adults were intranasally inoculated with a pre-Alpha SARS-CoV-2 virus strain. Extensive screening excluded participants with severe disease risk factors or comorbidities. Participants received the lowest quantifiable inoculum dose, resulting in no serious adverse events and resolved symptoms.
The study analyzed local and systemic immune responses at single-cell resolution. Baseline measurements were taken before inoculation, followed by time series analyses of cellular responses in blood and nasopharynx. Six participants developed sustained infections, defined by consecutive viral load detections and symptoms. Three individuals had sporadic positive polymer chain reaction (PCR) tests and were classified as transient infections. Seven participants remained PCR-negative but showed early innate immune responses, termed abortive infections. The infection rate was comparable to that in a closed household of unvaccinated individuals.
scRNA-seq and single-cell T cell receptor (TCR) and B cell receptor (BCR) sequencing were performed at up to seven time points. Cellular indexing of transcriptomes and epitopes by sequencing (CITE-seq) quantified 123 surface proteins in PBMCs. More than 600,000 single-cell transcriptomes were generated, including 371,892 PBMCs and 234,182 nasopharyngeal cells. Predictive models and marker gene expression annotated 202 cell states, enabling detailed local and systemic response analysis.
Generalized linear mixed models (GLMMs) quantified changes in cell-type abundance over time. Immune cell types infiltrated the inoculation site after SARS-CoV-2 exposure. Sustained infections showed immune infiltration starting at day 5, while transient infections had immediate infiltration at day 1. Abortive infections showed minimal changes except for early Cluster of Differentiation (CD)4+ and CD8+ T cell infiltration.
Gene expression analysis revealed that interferon response genes were the dominant infection-induced module in sustained infections. Interferon signaling was activated in all cell types in both blood and nasopharynx, peaking earlier in blood. This rapid systemic response was validated with bulk RNA-seq data. Myeloid cell redistribution between circulation and tissues was observed during early infection. Mucosal-Associated Invariant T (MAIT) cell activation was detected across all infection groups, indicating rapid viral sensing. Viral RNA peaked at day 7, with hyperinfected ciliated cells identified as major virion producers.
Ciliated cells showed dynamic responses, including acute-phase and interferon-stimulated states. Activated T cells, identified through peptide-Major Histocompatibility Complex (MHC) staining and scRNA-seq, expanded significantly at day 10 after inoculation, resembling a typical antigen-specific adaptive immune response.
Conclusions
The study revealed multiple immune response states that precede clinical symptoms, including MAIT cell activation and a decrease in inflammatory monocytes. These responses emerged even when SARS-CoV-2 exposure did not lead to COVID-19, suggesting their potential as biomarkers of immediate immune response. In sustained infections, a new acute phase response (APR) in ciliated cells and a distinct state for activated T cells with SARS-CoV-2-specific TCRs were identified. Interferon signaling was activated globally in circulating immune cells before the site of infection. These findings provide a detailed time-resolved description of early immune responses to SARS-CoV-2.