A sweeping review shows how the brain’s immune defenses can both fight and fuel neurodegeneration, revealing why timing, cell type, and disease context may be crucial for future therapies.
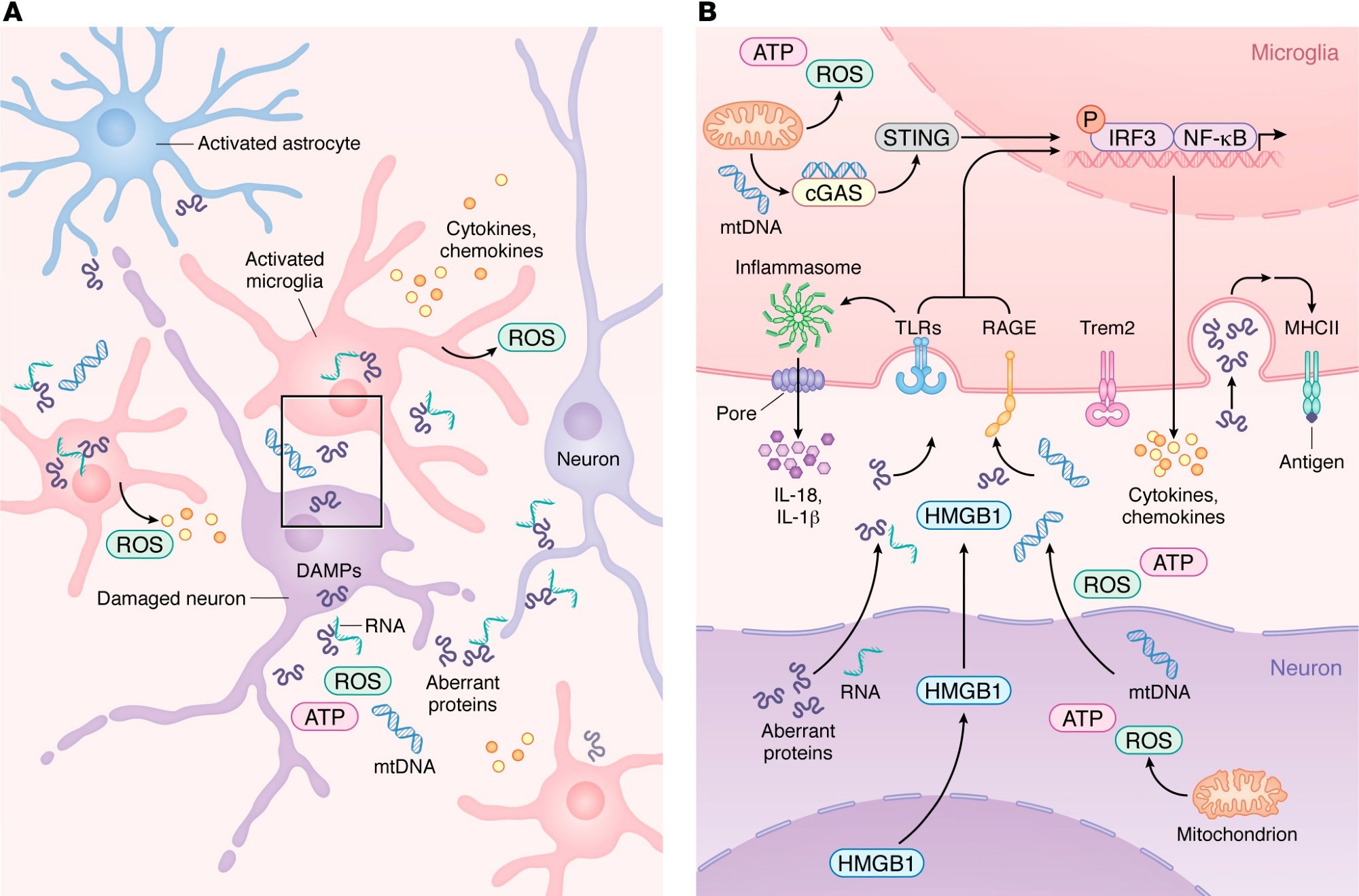
Innate immune activation and microglial transition during neurodegeneration. (A) During neurodegeneration, distressed neurons release DAMPs such as mtDNA, ROS, HMGB1, aberrant proteins, and pTau–bound RNA. These signals activate resident microglia and astrocytes, which then recruit peripheral immune cells. As shown in more detail in B, activated microglia upregulate PRRs, including TLRs, RAGE, and cGAS, enabling detection of neuron-derived DAMPs. Engagement of PRRs induces proinflammatory gene expression programs and activates inflammasome pathways that promote release of chemokines and cytokines into the CNS parenchyma. As microglia transition from homeostatic to reactive states in a TREM2-dependent manner, damage-associated microglia increase MHC-II expression to support antigen presentation and coordination of adaptive immune responses that attempt to limit neurodegeneration.
A recent review published in the Journal of Clinical Investigation synthesized current evidence on immune mechanisms in neurodegeneration. Accumulating evidence suggests that the immune system not only drives the progression of neurodegenerative diseases but also contributes to their onset through maladaptive cellular signaling and activation. The central nervous system (CNS) parenchyma depends on resident immune cells to maintain the blood-brain barrier (BBB) and respond to dysfunctional cells, pathogens, or cellular damage under steady-state conditions.
Neurons activate signaling pathways to respond to cellular dysfunction, while microglia and astrocytes alleviate danger signals and recruit peripheral immune cells. However, these responses can aggravate aberrant protein deposition during neurodegeneration, and the balance between detrimental and beneficial immune involvement is poorly defined. In the present review, researchers reviewed current knowledge of immune mechanisms in neurodegeneration.
Innate Immune Signaling in Neurodegenerative Disease
Aberrant proteins, including phosphorylated tau, amyloid β (Aβ), and α-synuclein, have been identified as damage-associated molecular patterns (DAMPs) that activate pattern recognition receptors in the CNS. Toll-like receptor 2 (TLR2) and TLR4 are upregulated in mouse models of Alzheimer’s disease (AD) and in the brains of AD patients.
The binding of Aβ to TLR4 or TLR2 elevates the expression of cytokines, including tumor necrosis factor (TNF)-α, interleukin (IL)-6, IL-1β, IL-17, and IL-10, in microglia and astrocytes. Pharmacological inhibition or genetic ablation of these TLRs worsens cognitive decline and increases Aβ burden in the brain by decreasing microglial activation. The review also highlighted inflammasome and DNA-sensing pathways, including NLRP3 and cGAS-STING, as additional contributors to neuroinflammation across neurodegenerative diseases.
Studies have consistently reported increased expression of the receptor for advanced glycation end products (RAGE) on neurons, astrocytes, and microglia in AD, Parkinson’s disease (PD), and amyotrophic lateral sclerosis. RAGE overexpression in mouse AD models accelerated cognitive impairment, while its deficiency decreased memory deficits.
Genetic studies in AD patients identified two single-nucleotide polymorphisms (SNPs) in the triggering receptor expressed on myeloid cells 2 (TREM2), which is highly expressed by microglia, as risk factors of a magnitude comparable to carrying the apolipoprotein E (APOE) ε4 allele. These observations indicate that microglia are key players in neurodegenerative pathogenesis.
T Cell Responses Across Neurodegenerative Disorders
A growing body of evidence indicates that cluster of differentiation 4 (CD4+) T cells have disease-specific functions across neurodegenerative diseases. Aβ-restricted CD4+ T cell transfusion into a transgenic AD mouse model was neuroprotective, preventing cognitive decline. In contrast, α-synuclein-specific CD4+ T cells were neurotoxic in PD models, promoting inflammation.
α-synuclein overexpression increased recruitment of CD4+ T cells producing interferon-γ, while depleting these cells decreased neuronal loss. These observations indicate that CD4+ T cells can be protective in some neurodegenerative diseases and pathogenic in others. In addition, CD8+ T cells have garnered increasing attention as contributors to CNS pathology. CD3+ and CD8+ T cells have been detected in the hippocampus in AD, with CD3+ T cell numbers correlating with tau pathology.
Similarly, increased CD8+ T cell numbers were observed in PD and correlated with neuronal death, although whether this relationship is causative or reactive remains uncertain. Further, the accumulation of neuroprotective CD8+ T cells in the brains of AD mice has been reported and shown to restrict amyloid plaque growth. Overall, studies reveal that CD8+ T cells have diverse roles in neurodegenerative diseases, shaped by antigen specificity, microglial interactions, and tissue residency. The review also noted growing interest in granzyme K-expressing CD8+ T cells as potentially specialized players in several neurodegenerative conditions.
Aging, Injury, and Viral Effects on CNS Immunity
Aging is a major risk factor for neurodegenerative diseases. In youth, microglia survey the CNS, astrocytes maintain the BBB, and only a small number of T cells enter the parenchyma. However, in advanced ages, chronic low-grade inflammation occurs, during which many immune pathways become dysregulated.
Further, repetitive head injury can accelerate neurodegeneration. Traumatic brain injury is linked to the development of PD, AD, and chronic traumatic encephalopathy, even when it occurs in childhood. Following mild traumatic brain injury, the release of DAMPs by distressed cells triggers microglial activation, which clears debris and reinforces weakened BBB regions.
Repetitive injuries may increase reactivity and trigger microglial death, thereby allowing the entry of myelomonocytic cells. Like repetitive injuries, viral infections can alter CNS immunity and influence vulnerability to neurodegeneration. Recent studies have linked non-neurotropic and neurotropic viral infections to a higher risk of neurodegenerative diseases.
Immune Timing and Neurodegeneration Implications
Taken together, neurodegenerative diseases stem from a complex interplay of environmental and biological factors that govern the magnitude and timing of immune activation and the programming of T cells, microglia, and other leukocytes. Viral infection, repetitive head injury, and aging restructure the CNS immune environment. Many immune pathways involved exhibit stage-dependent and context-specific functions.
Microglial activation may initially help clear aberrant proteins, but could become maladaptive with protracted stimulation. Similarly, CD8+ T cells can promote neuronal injury or support tissue repair based on antigen specificity, local signaling cues, and effector programming. As such, appreciating the cell-intrinsic states and temporal dynamics of immune cells is crucial for understanding immune signatures and determining when immunomodulatory interventions will be beneficial.