Since late 2021, the SARS-CoV-2 Omicron variant consistently caused a majority of infections, with over three novel subvariants that could effectively evade immunity. In the spring of 2022, an early wave of infections due to the Omicron BA.1 subvariant was rapidly displaced by a wave of the BA.2 subvariant, which was subsequently replaced by the BA.5 subvariant. The BA.5 was significantly more contagious and accounted for 75% to 95% of coronavirus disease 2019 (COVID-19) cases by August 2022.
Since a significant proportion of the global population was infected with Omicron at the beginning of 2022, research is centered on whether convalescent individuals acquired natural immunity that could protect them from BA.4/BA.5 subvariants. Serum neutralization studies have shown that neither immunization nor previous infection diagnosed during the early waves of Omicron infections provided sufficient protection against BA.5. Recent studies have revealed that a comparatively lesser number of BA.5-infected patients were previously diagnosed with Omicron infection, suggesting that prior Omicron infection could protect against BA.5 infection
About the study
In the present study, researchers examined authentic viral neutralization capacity between those who received three-dose BNT162b2 vaccination regimens to those who were immunized and later infected with SARS-CoV-2 Omicron BA.1/BA.2 subvariants.
In Odense, Denmark, the team recruited healthy individuals from the Odense University Hospital personnel and the general public. Four weeks after receiving their third BNT162b2 vaccination, serum samples were collected from the 24 members of the vaccinated cohort between 18 November 2021 and 4 February 2022. Additionally, serum samples were collected from 12 individuals from the convalescent cohort between 26 January and 19 April 2022, four weeks after the participants reported Omicron BA.1/BA.2 breakthrough infections.
Plaque reduction neutralization tests (PRNTs) were conducted on authentic SARS-CoV-2 Delta and Omicron BA.1, BA.2, and Ba.5 clinical isolates. The team measured PRNT90 titers, the highest serum dilution resulting in more than 90% plaque reduction. Lineages were identified via nanopore whole-genome sequencing (WGS). All serum samples were tested for spike-specific antibodies. In addition, using the Alinity SARS-CoV-2 immunoglobulin (Ig)-G assay, serum samples were evaluated for nucleocapsid-specific antibodies to determine SARS-CoV-2–naive status within the vaccinated group.
Results
Serum samples from the vaccinated cohort showed neutralization of Omicron BA.5 subvariant at a median PRNT90 titer of 20. In contrast, serum samples of the convalescent cohort displayed BA.5 neutralization at a median PRNT90 titer of 160. Furthermore, Wilcoxon rank-sum test indicated that this eight-fold rise in neutralization following Omicron infection had statistical significance. Additionally, the convalescent cohort displayed neutralization of SARS-CoV-2 Delta and Omicron BA.1/BA.2 subvariants at considerably higher levels than the vaccinated cohort. In the convalescent and vaccinated groups, BA.5 neutralization was noted at median titers that were two to eight times lesser than the other viral strains.
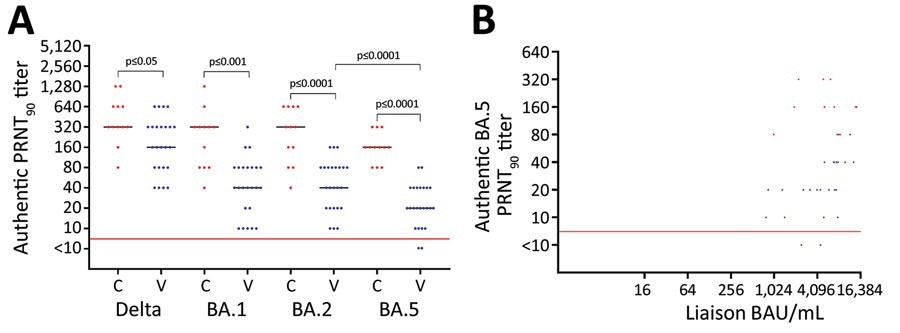
Neutralization of SARS-CoV-2 variants among vaccine-boosted persons with and without prior Omicron BA.1/BA.2 infections, Denmark. A) PRNT90 titers against SARS-CoV-2 Delta variant and Omicron variants BA.1, BA.2, and BA.5. B) Correlation between the levels of spike antibodies and PRNT90 titers. Participants received 3 doses of BNT162b2 (Pfizer-BioNTech), 2 initial vaccines and a booster dose. We analyzed titers for 24 vaccinated participants (blue dots) who received 3 BNT162b2 doses only and 12 convalescent participants (red dots) who received 3 vaccine doses and had Omicron BA.1/BA.2 infection. For statistical analysis, a Kruskal-Wallis test was applied initially to account for the multiple comparisons problem. Subsequently, unpaired PRNT90 titers were compared with the Wilcoxon rank-sum test, whereas paired PRNT90 titers were compared with the Wilcoxon sign rank test. Red horizontal lines indicate neutralization threshold; horizontal bars indicate median neutralization titer for each SARS-CoV-2 strain. BAU, binding antibody units; C, convalescent participant; PRNT90, plaque reduction neutralization tests with plaque reduction >90%; V, vaccinated participant.
The median concentrations of SARS-CoV-2 spike-specific antibodies varied between cohorts, with the vaccinated and convalescent cohorts having 5,535 and 5,675 binding antibody units/mL, respectively. However, the Wilcoxon rank-sum test revealed that this variation was not statistically important. In addition, an association was observed between the concentrations of viral spike antibodies and PRNT90 titers within the vaccinated cohort, while a similar relationship was not found in the convalescent group.
According to in vitro neutralization investigations, past BA.1/BA.2 infection confers no discernible immunity against BA.5. The team evaluated two small but closely matched cohorts with comparable demographics, blood sampling intervals, and antibody levels. Infections diagnosed in the Omicron BA.1/BA.2 wave significantly improved BA.5 neutralizing capacity in those receiving three-dose vaccination regimens.
Overall, the study showed that infection with Omicron BA.1/BA.2 appeared to lower susceptibility to emerging Omicron subvariants like BA.5 in individuals who received three doses of the BNT162b2 vaccination. In conjunction with the newly reported slower decline of Omicron infection-induced immunity, the study findings suggest that prior Omicron infection significantly increases humoral protection.
Journal reference:
- Pedersen RM, Bang LL, Tornby DS, Madsen LW, Holm DK, Syndenham TV, et al., Omicron BA.5 neutralization among vaccine-boosted persons with prior Omicron BA.1/BA.2 infections, Emerg Infect Dis. 2022 Dec [date cited], DOI: https://doi.org/10.3201/eid2812.221304, https://wwwnc.cdc.gov/eid/article/28/12/22-1304_article