AptaFluor SAH Methyltransferase Assay is a high-throughput enzymatic assay for SAM-dependent methyltransferases. The assay uses a naturally existing SAH-sensing RNA aptamer, or riboswitch, which binds SAH with nanomolar affinity and excellent selectivity.
These split-aptamer sensors produce positive time-resolved Förster resonance energy transfer (TR-FRET) signals after binding to SAH concentrations as low as 1 nM. Consequently, the AptaFluor SAH Methyltransferase Assay overcomes the technical limitations of conventional detection techniques, which only detect certain methylation events or are insensitive to enzymes with low turnovers or high SAM affinities.
This article shows how the Aptafluor SAH assay can offer a robust and dependable tool for measuring the activity of four methyltransferases (MTs) using a variety of acceptor substrates, such as peptides, DNA, and RNA. Selected enzymes with typically low Km (100 nM to 5 µM) have attracted significant interest as therapeutic targets. Among these enzymes, PRMT5 and MLL4 are histone methyltransferases that catalyze arginine and lysine methylation, respectively. METTL3/14 methylates adenosine at the N6 position (m6A) in mRNA substrates with a DRACH motif, whereas NSP14 catalyzes the methylation of viral RNAs at N7-guanosine during the cap formation process.
The assay’s selectivity, sensitivity, reagent stability, and tolerance to several acceptor substrates are demonstrated. Although the assay uses an RNA aptamer, it also tolerates dsRNAs and some ssRNAs. The assay allows reliable detection of SAH (Z’ > 0.7) at enzyme concentrations as low as nM, and can be used to profile dose-response curves for known inhibitors.
AptaFluor SAH Methyltransferase Assay: Direct, homogenous SAH detection with a TR-FRET readout
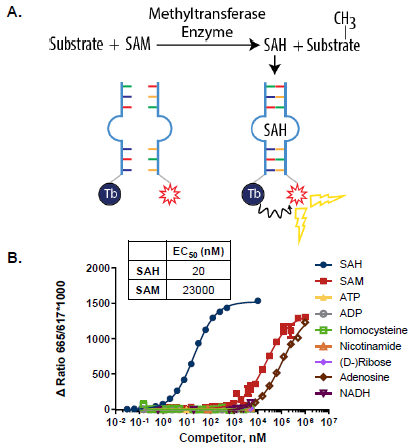
Figure 1. AptaFluor SAH Methyltransferase Assay Principle: A. SAH-driven assembly of a split aptamer allows FRET between a Terbium chelate donor and a DyLight 650 acceptor. B. Selectivity analysis. The modified SAH riboswitch discriminates between SAH and related nucleotides, including SAM, which differs by a single methyl group. This makes unambiguous detection of SAH possible in the presence of excess SAM. Image Credit: BellBrook Labs
Sensitivity and reagent stability
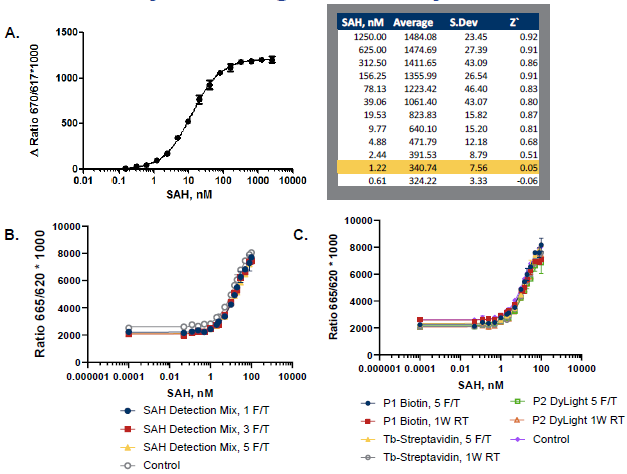
Figure 2. Assay Sensitivity and Reagent Stability. A. Excellent Z` and lower limit of detection obtained using the AptaFluor assay. B-C. Reagent stability was assessed using 100 nM SAM/SAH standard curves after indicated freeze-thaw (F/T) cycles or storage conditions. The tests were performed using (B) the combined detection reagents (SAH Detection Mix) or (C) individual reagents (P1 Biotin, TbStreptavidin, or P2 DyLight), n=2. Image Credit: BellBrook Labs
Universal MT detection with any acceptor
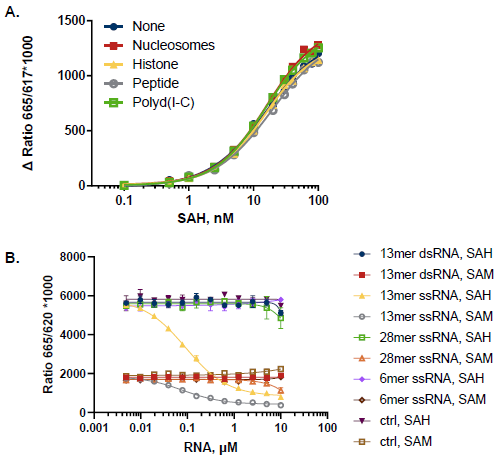
Figure 3. Toleration of MT Acceptor Substrates. A. 100 nM SAH-SAM standard curves were set up in the presence of nucleosomes (10 ng/µL), histone H3-3 (3 ng/µL), Histone H3 (1-21) peptide (10 µM), and Polyd(I-C) (2.5 mU/µL); control wells lacked a MT substrate. B. RNA titrations at 100 nM SAH or 100 nM SAM; control wells lacked RNAs. dsRNAs do not interfere with the assay while some ssRNA sequences interfere with the detection. Image Credit: BellBrook Labs
PRMT5 enzyme activity
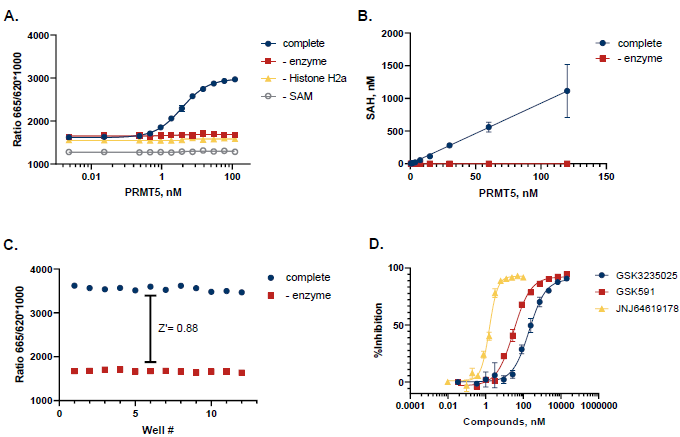
Figure 4. Case Study for PRMT5. A. PRMT5 was titrated in the presence of 5 µM SAM and 5 µM Histone H2a. Reactions were incubated at 30°C for 90 minutes. B. TR-FRET values were converted to SAH production to assess reaction progress. C. A Z’ of 0.88 was obtained (n=12) for an initial velocity reaction (10 nM PRMT5, 90 min). D. Dose response curves with known probe inhibitors GSK3235025, GSK591, and JNJ64619178 showed IC50 values of 207 nM, 30.7 nM, and 1.59 nM, respectively. Image Credit: BellBrook Labs
MLL4 enzyme activity
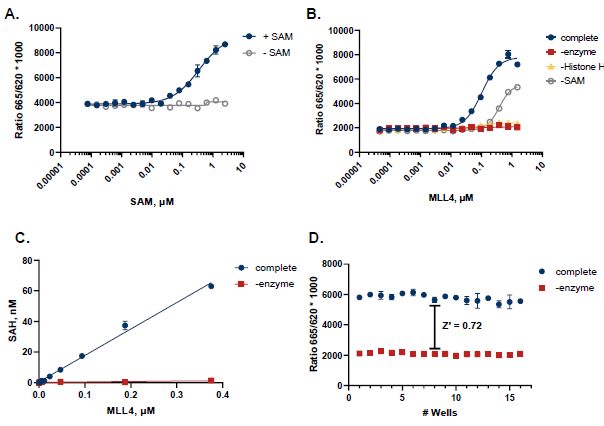
Figure 5. Case Study for MLL4. A. SAM titration showed apparent Km at 350 nM B. MLL4 was titrated in the presence of 350 nM SAM and 10 µM Histone H3. Reactions were incubated at 30 °C for 2 hours. C. TR-FRET values were converted to SAH production to assess reaction progress. D. A Z’ of 0.72 was obtained (n=16) for an initial velocity reaction (250 nM MLL4, 2 hours). Image Credit: BellBrook Labs
METTL3/METTL14 enzyme activity
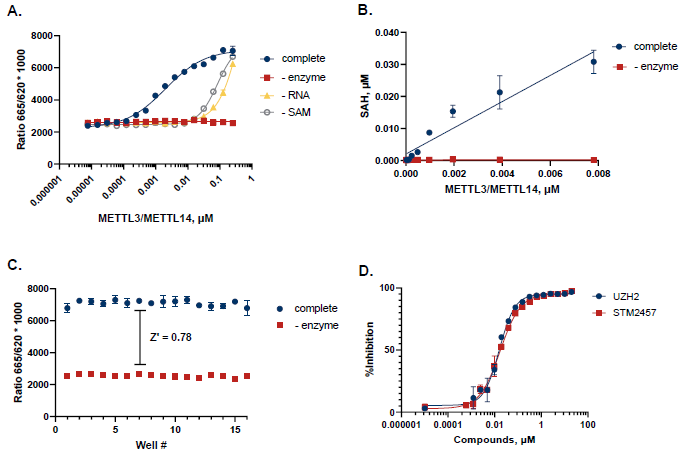
Figure 6. Case Study for METTL3/METTL14. A. METTL3/METTL14 was titrated in the presence of 250 nM SAM and 1 µM ssRNA Oligomer. The sequence of the ssRNA is 5’- GUUGCCUGUUCGUGUUGGACUUGCCUGU-3’. Reactions were incubated at 30°C for 2 hours. B. TR-FRET values were converted to SAH production to assess reaction progress. C. A Z’ of 0.78 was obtained (n=16) for an initial velocity reaction (7.5 nM METTL3/METTL14, 2 hours). D. Dose response curves with known probe inhibitors UZH2 and STM2457 showed IC50 values of 16 nM and 17 nM, respectively. Image Credit: BellBrook Labs
NSP14 enzyme activity
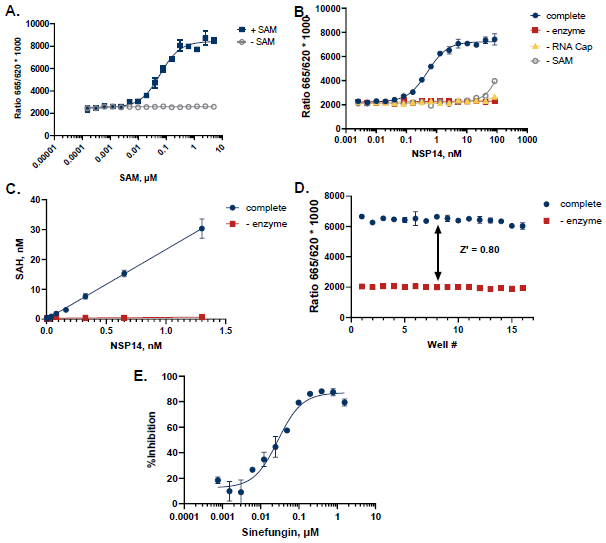
Figure 7. Case Study for NSP14. A. SAM titration showed apparent Km at 60 nM B. NSP14 was titrated in the presence of 100 nM SAM and 50 µM of a G(5')ppp(5')A RNA Cap Structure Analog. Reactions were incubated at 37 °C for 90 minutes. C. TR-FRET values were converted to SAH production to assess reaction progress. D. A Z’ of 0.80 was obtained (n=16) for an initial velocity reaction (1.45 nM NSP14, 90 min). E. Dose response curves with known probe inhibitor Sinefungin showed IC50 values of 28 nM. Image Credit: BellBrook Labs
Conclusions
- The AptaFluor SAH Methyltransferase Assay utilizes the excellent affinity and selectivity of a microbial riboswitch for direct identification of SAH with a TR-FRET signal.
- Ultra-Sensitive and Broad Dynamic Range - the assay has a LLD of approximately 1 nM SAH, allowing the detection of MTs with SAM concentrations as low as 100 nM and exceptional Z’ values.
- Excellent Reagent Stability - reagents are stable after several freeze-thaw cycles and at room temperature, allowing flexibility to the user.
- Universal - tolerates conventional acceptor substrates, from peptides to RNAs or DNAs, and allows detection of different MTases, such as histone, RNA, and DNA.
About BellBrook Labs
BellBrook Labs is dedicated to providing scientists with enabling screening tools to accelerate the discovery of more effective therapies. Leveraging its two base platforms, BellBrook has developed easy-to-use assays for hundreds of drug targets.
Transcreener® Biochemical Assay Technology
The proprietary Transcreener HTS Platform uses a highly specific antibody and far-red tracer for fluorescent immunodetection of nucleotides, including ADP, UDP, GDP, AMP, and GMP. Because its based on detection of nucleotides, the assay is universal for use with virtually any enzyme that produces these nucleotides, such as kinases, glycosyltransferases, GTPases, helicases, ATPases, nucleotidases, exonucleases and PDEs. The assay boasts direct detection of many of these enzyme targets (no coupling enzyme needed), simplyifing the protocol and reducing compound interference.
AptaFluor® Biochemical Assay Technology
AptaFluor leverages a spit aptamer technology to directly detect SAH, the common product of Methyltransferases. As the most sensitive HTS methyltransferase activity assay available, AptaFluor dramatically reduces enzyme usage and allows the assay to be run at or below Km for SAM.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.